ISSN: 1839-9940
J Genomics 2023; 11:26-36. doi:10.7150/jgen.81013 This volume Cite
Research Paper
Draft Genome Sequences of Synechococcus sp. strains CCAP1479/9, CCAP1479/10, CCAP1479/13, CCY0621, and CCY9618: Five Freshwater Syn/Pro Clade Picocyanobacteria
1. School of Geographical Sciences, Faculty of Science, University of Bristol, Bristol, BS8 1SS, United Kingdom.
2. CNRS and Sorbonne Université, FR 2424, Roscoff Culture Collection, Station Biologique de Roscoff (SBR), Roscoff, France
3. Department of Marine Microbiology and Biogeochemistry, Royal Netherlands Institute for Sea Research, Den Hoorn, the Netherlands.
Received 2022-11-18; Accepted 2023-3-13; Published 2023-4-25
Abstract
Picocyanobacteria are essential primary producers in freshwaters yet little is known about their genomic diversity and ecological niches. We report here five draft genomes of freshwater picocyanobacteria: Synechococcus sp. CCAP1479/9, Synechococcus sp. CCAP1479/10, and Synechococcus sp. CCAP1479/13 isolated from Lake Windermere in the Lake District, UK; and Synechococcus sp. CCY0621 and Synechococcus sp. CCY9618 isolated from lakes in The Netherlands. Phylogenetic analysis reveals all five strains belonging to sub-cluster 5.2 of the Synechococcus and Prochlorococcus clade of Cyanobacteria. These five strains are divergent from Synechococcus elongatus, an often-used model for freshwater Synechococcus. Functional annotation revealed significant differences in the number of genes involved in the transport and metabolism of several macro-molecules between freshwater picocyanobacteria from sub-cluster 5.2 and Synechococcus elongatus, including amino acids, lipids, and carbohydrates. Comparative genomic analysis identified further differences in the presence of photosynthesis-associated proteins while gene neighbourhood comparisons revealed alternative structures of the nitrate assimilation operon nirA.
Keywords: Freshwater, Picocyanobacteria, Synechococcus, Genome, Synechococcus sp. CCAP1479/9, Synechococcus sp. CCAP1479/10, Synechococcus sp. CCAP1479/13, Synechococcus sp. CCY0621, Synechococcus sp. CCY9618
Introduction
Picocyanobacteria play a key role in aquatic ecosystems, contributing a significant proportion of total primary production in both marine and fresh waters [1-3]. These unicellular cyanobacteria, sized between 0.5 and 2 μm, are distributed globally, from temperate and tropical open oceans to alpine lakes and eutrophic reservoirs [4-6]. Freshwater picocyanobacteria are predominantly Synechococcus strains which can dominate the picophytoplankton component (1 - 99% [7]) and total biomass (10 - 70% [8]) depending on trophic status and depth [9,10]. Other taxonomic names associated with freshwater picocyanobacterial strains are Cyanobium spp. [11] and Vulcanococcus spp. [12].
The availability of sequenced freshwater picocyanobacteria genomes has lagged behind that of marine picocyanobacteria (Prochlorococcus and Synechococcus) [13]. This has limited genomic approaches to understand freshwater picocyanobacteria with regards to ecology and evolution - a hot topic in both freshwater and marine environments [14-18]. A further limitation is the divergence seen among freshwater Synechococcus clades. Though Synechococcus elongatus cells are larger than those of the Syn/Pro clade Synechococcus [19,20], and do not fall under the 'pico-' threshold, they are often used as models for freshwater picocyanobacteria [21-25]. However, the emergence of the Synechococcus elongatus strains as a deep branching sister group to the monophyletic Syn/Pro clade suggests Synechococcus elongatus provides an unrepresentative model of freshwater picocyanobacteria and freshwater Synechococcus [26]. Freshwater strains of the Syn/Pro clade have a wider geographic distribution than Synechococcus elongatus and may have a greater ecological influence [27], yet their molecular capabilities are poorly understood in comparison to Synechococcus elongatus. Here, we have sequenced draft genomes of five new picocyanobacteria to increase genomic representation of the freshwater strains in the Syn/Pro clade. Three were isolated from Lake Windermere in the UK: Synechococcus sp. CCAP1479/9, Synechococcus sp. CCAP1479/10, and Synechococcus sp. CCAP1479/13. The remaining two were isolated from ponds in the Netherlands: Synechococcus sp. CCY0621 (Leiden) and Synechococcus sp. CCY9618 (Vinkeveen).
Materials and Methods
Three Synechococcus strains were obtained from the Culture Collection of Algae and Protozoa: Synechococcus sp. CCAP1479/9, Synechococcus sp. CCAP1479/10, and Synechococcus sp. CCAP1479/13, all isolated from Lake Windermere, UK. Two Synechococcus strains were obtained from the Culture Collection Yerseke: Synechococcus sp. CCY0621 and Synechococcus sp. CCY9618, isolated from ponds in The Netherlands (Leiden and Vinkeveen respectively) (Supplementary Figure S1). All strains were grown in BG-11 medium [28] at 20 °C with 10-20 µmol m-2 s-1 of white light under a 16 h: 8 h light:dark cycle.
Aliquots of 1.8 mL of each mono-phototrophic culture were harvested to extract genomic DNA using DNeasy UltraClean Microbial Kits (Qiagen, Germany) according to the manufacturer's instructions. Once purified, genomic DNA was stored at -80 °C in 10 mM Tris buffer at pH 8. DNA concentration and quality was measured using a NanoDrop 2000 spectrophotometer (Thermo Scientific, USA) and a Qubit 2.0 Fluorometer (Thermo Scientific, USA).
Whole genome library preparation and sequencing was carried out by the University of Bristol Genomics Facility, UK. DNA libraries were prepared for each strain using Truseq Nano LT Kit (Illumina, USA) and sequenced using Illumina NextSeq 500/550 Mid Output Kit v2 (300 cycles) (Illumina, USA) to generate paired-end reads (2 x 150 bps). Raw reads were trimmed using Trimmomatic v0.39 [29] with parameters Leading: 20, Trailing: 20, SlidingWindow: 4:20, MinLen: 20, and assembled de novo using SPAdes v3.14.1 [30] with k-mers of 67, 77, 87, 97 and a coverage cutoff of 20 in --careful mode. A BLAST database was generated at the amino acid sequence level for each assembly and searched against a collection of 1,054 core cyanobacterial genes (CCGs) [31,32]. Bandage v0.8.1 [33] was used to visualise strain assemblies and separate out cyanobacterial sequences based on contiguous CCG-containing nodes as demonstrated in previous assemblies [32]. Contigs which did not contain cyanobacterial genes were discarded, in addition to short (<200 bp) contigs. The assembled genomes had overall coverages ranging from 552x to 939x (Table 1) and structurally annotated with GeneMark.hmm-2 v1.05 [34], Prodigal v2.6.3 [35], INFERNAL v1.1.2 [36], and tRNAscan-SE v2.0.5 [37]. Genome completeness was estimated by identifying cyanobacteria-specific single-copy orthologous genes using BUSCO v3.0.2 [38]. The draft genomes were submitted to JGI IMG/ER [39] (GOLD Analysis Project IDs: Ga0436386, Ga0436387, Ga0436388, Ga0436389, and Ga0436390). The five draft genomes were deposited to the DDBJ/Genbank/ENA repositories with accession numbers JAFKRG000000000 (CCY9618), JAFKRH000000000 (CCY0621), JAFKRI000000000 (CCAP1479/13), JAFKRJ000000000 (CCAP1479/10), and JAFKRK000000000 (CCAP1479/9).
Genomic features of the sequenced freshwater picocyanobacteria
| Synechococcus sp. CCAP1479/9 | Synechococcus sp. CCAP1479/10 | Synechococcus sp. CCAP1479/13 | Synechococcus sp. CCY0621 | Synechococcus sp. CCY9618 | |
|---|---|---|---|---|---|
| Genome size (bp) | 3,288,920 | 3,313,705 | 3,299,582 | 3,230,971 | 2,927,161 |
| Contigs | 88 | 108 | 132 | 101 | 133 |
| N50 (bp) | 207,208 | 151,487 | 78,719 | 105,719 | 94,487 |
| Genome coverage | 825X | 939X | 552X | 818X | 865X |
| DNA coding (%) | 91.98 | 91.99 | 91.92 | 90.96 | 90.5 |
| DNA G+C (%) | 69.36 | 69.33 | 69.32 | 67.45 | 68.34 |
| Total genes | 3,423 | 3,502 | 3,507 | 3,471 | 3,165 |
| Protein encoding genes | 3,364 | 3,441 | 3,446 | 3,407 | 3,109 |
| Completeness (%) | 98.4 | 98.7 | 98.2 | 98.6 | 98.6 |
| Average Nucleotide Identity to Synechococcus elongatus PCC 7942 | 73.5808 | 73.4985 | 73.5014 | 73.3739 | 73.3276 |
Functional annotation was determined through the eggNOG web server [40]. Two-tailed t-tests were applied to carry out statistical analysis on total COG numbers and COGs normalised as a proportion of total genome. JGI IMG/ER was used to carry out KEGG [41] comparative genomic analysis for photosynthesis and nitrate metabolism pathways between Synechococcus elongatus (Synechococcus elongatus PCC 7942, Synechococcus elongatus UTEX 2973, Synechococcus elongatus PCC 6301, Synechococcus elongatus FACHB-242, Synechococcus elongatus FACHB-1061) and the sequenced Synechococcus strains.
The evolutionary relationships of the newly sequenced strains with a selection of cyanobacterial taxa sampling a broad range of morphologies, lifestyles, and metabolisms, were estimated through phylogenetic analysis. Our dataset included 373 cyanobacteria genomes and ortholog sequences from 143 protein-coding genes, based on previously published studies [42-44]. We performed BLAST searches with these ortholog sequences against the 373 genomes using blastp v2.11.0+ [45] with an e-value threshold of 10-5, retaining the hit with the highest score and extracting the corresponding protein sequences. The resulting sequences were aligned using MAFFT v7.511 [46] with the -localpair -maxiterate 1000 parameters. Maximum-likelihood gene trees were constructed using IQ-TREE 2.2.0 [47], implementing the LG protein evolution model and the -fast option. These gene trees were used to identify the clusters of sequences that were most closely associated with the BLAST query sequences - these clusters were assumed to be 'true' orthologs. These true orthologs were re-aligned with MAFFT (same parameters as above) and inspected with mis-aligned columns and alignment positions with a gap content higher than 80% removed from each alignment. The best evolutionary model for each gene was determined by using IQ-TREE with the -m MF option [48], selecting the model with the lowest BIC score. A maximum-likelihood partitioned phylogenetic analysis was performed using IQ-TREE [49]. Using the previously determined evolutionary models, partitioned analysis was carried out with IQ-TREE using -p and -B 1000 parameters with each gene assigned to its own partition. The -p option constrains all partitions to the same topology and branch length but allows each partition to have a different overall evolutionary rate, while -B 1000 produces ultrafast bootstrap support values [50]. This analysis was carried out twice with the two resulting trees compared to confirm no significant differences between them.
Results and Discussion
The newly sequenced picocyanobacteria genomes consist of 88 to 133 contigs (average of 112) and range in size from 2.9 Mbps to 3.3 Mbps (average of 3.2 Mbps), significantly larger than Synechococcus elongatus strains (p < .001, n = 5). Synechococcus sp. CCY9618 has the smallest genome and is composed of the largest number of contigs with an N50 value of 94,487 (Table 1). Synechococcus sp. CCAP1479/10 has the largest genome, while Synechococcus sp. CCAP1479/9 contains the fewest contigs (88) and the largest N50 (207,208). Genome coverage is high among the assemblies (552x - 939x) with genome completeness estimated at 98.2 - 98.7%. It should be noted that these genomes have not been completely closed yet a high genome completeness suggests that the 'missing' part of the genome is limited.
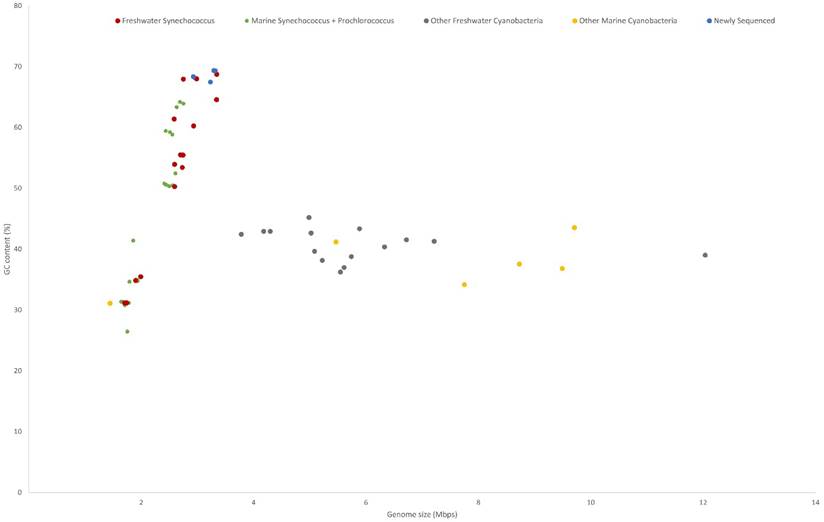
All five genomes contain high GC contents ranging from 67.45 - 69.36% (Table 1). This is consistent with previously sequenced freshwater picocyanobacteria and Synechococcus elongatus, regularly featuring a high (>60%) GC content [13,18]. Compared to marine Syn/Pro strains, freshwater Synechococcus have significantly larger genome sizes (p < .001, n = 20; primarily due to genomic streamlining of Prochlorococcus spp. [51]) and higher GC content (p < .001, n = 20) (Figure 1). Meanwhile, the trend of increasing GC content with increasing genome size present in freshwater and marine Synechococcus is not found in larger cyanobacteria (cell size greater than 2 µm). Higher genomic GC contents have been linked with increased horizonal gene transfer and protection against DNA damage through higher resilience against UV irradiation, contributing to picocyanobacterial genomic plasticity and environmental adaptability [52,53]. Conversely, lower GC contents in marine picocyanobacteria may indicate selection in N limited environments due to the reduced N requirement for AT pairs [54].
GC content and genome size of cyanobacteria characterised into habitat and phylogeny. Freshwater Synechococcus include picocyanobacteria found in the Syn/Pro clade and Synechococcus elongatus strains. The newly sequenced strains are clustered with the freshwater Synechococcus.
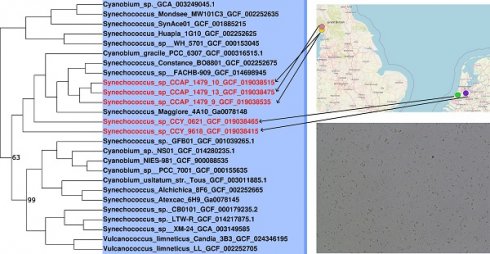
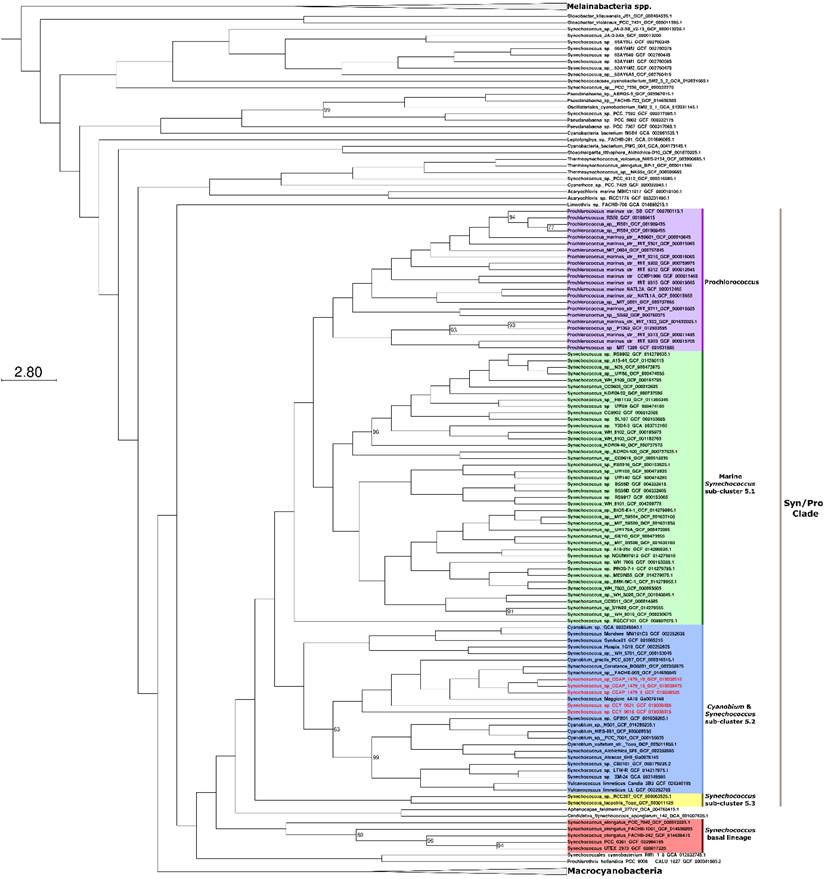
Phylogenomic analyses were carried out to identify the closest relatives of the newly sequenced freshwater picocyanobacteria. All five strains belong to the Cyanobium and Synechococcus freshwater sub-cluster 5.2 of the Syn/Pro clade (Figure 2). Synechococcus sp. CCAP1479/10, Synechococcus sp. CCAP1479/13, and Synechococcus sp. CCAP1479/13 form a monophyletic clade, with Synechococcus sp. BO8801 (Lake Constance, Germany) and Synechococcus sp. FACHB-909 (Baohu Lake, China) the closest related strains (a sister group to these three newly sequenced picocyanobacteria). Synechococcus sp. CCY0621 and Synechococcus sp. CCY9618 are more distantly related and appear as outgroups to the CCAP newly sequenced strains. In contrast, Synechococcus elongatus strains are a sister group of the Syn/Pro.
Maximum likelihood phylogeny showing the relationship of Synechococcus sp. CCAP 1479/9, Synechococcus sp. CCAP 1479/10, Synechococcus sp. CCAP 1479/13, Synechococcus sp. CCY 0621, and Synechococcus sp. CCY 9618 within the Syn/Pro clade. Newly sequenced picocyanobacteria are highlighted in red. The tree was constructed from 373 cyanobacteria and 145 orthologous proteins. Bootstrap values less than 100 are displayed at branching nodes while blank nodes have a support of 100. The tree is rooted using Melainabacteria spp. as an outgroup. The scale bar represents an average of 2.8 substitutions per site. An expanded tree is shown in Supplementary Information Figure S2.
Freshwater picocyanobacteria from the Syn/Pro clade are derived taxa that specialised in a planktonic habitat. The newly sequenced genomes were functionally annotated with eggNOG and KEGG, in addition to five Synechococcus elongatus genomes (Synechococcus elongatus PCC 7942, Synechococcus elongatus UTEX 2973, Synechococcus elongatus PCC 6301, Synechococcus elongatus FACHB-242, and Synechococcus elongatus FACHB-1061). This enabled insights into the genomic capabilities of the scarcely researched freshwater sub-cluster 5.2 of the Syn/Pro clade compared to the Synechococcus elongatus basal lineage.
Of the 19 functional COG categories identified, 11 categories differed significantly between our sequenced genomes and Synechococcus elongatus strains, in terms of total gene number and genes as a percentage of the total genome (Table 2, Supplementary Table S1). Five of these categories were found to be significantly increased in our sequenced genomes (V, M, G, E, I), while three were significantly decreased (J, N, F). The total number of genes associated with three categories (O, C, H) were significantly greater in our sequenced genomes (p < .001, n = 5), though as a proportion of their genome were significantly greater in Synechococcus elongatus strains (p = .006, p = .007, p < .001 respectively, all n = 5). Additionally, KEGG analysis revealed 1,425 KO terms within at least one of the sub-cluster 5.2 freshwater picocyanobacteria of which 183 terms were not identified in Synechococcus elongatus strains. Meanwhile, 162 KO terms are found in Synechococcus elongatus but absent from our newly sequenced strains (Supplementary Table S2).
Number of eggNOG classifications of proteins encoded by the five sequenced sub-cluster 5.2 Synechococcus genomes and five selected Synechococcus elongatus strains. Percentage of genes as proportion of the genome is provided in brackets. J: Translation, ribosomal structure and biogenesis; K: Transcription; L: Replication, recombination and repair; B: Chromatin structure and dynamics; D: Cell cycle control, cell division, chromosome partitioning; V: Defence mechanisms; T: Signal transduction mechanisms; M: Cell wall/membrane/envelope biogenesis; N: Cell motility; U: Intracellular trafficking, secretion, and vesicular transport; O: Posttranslational modification, protein turnover, chaperones; C: Energy production and conversion; G: Carbohydrate transport and metabolism; E: Amino acid transport and metabolism; F: Nucleotide transport and metabolism; H: Coenzyme transport and metabolism; I: Lipid transport and metabolism; P: Inorganic ion transport and metabolism; Q: Secondary metabolites biosynthesis, transport and catabolism; S: Function unknown.
| COG | Synechococcus sp. CCAP1479/9 | Synechococcus sp. CCAP1479/10 | Synechococcus sp. CCAP1479/13 | Synechococcus sp. CCY0621 | Synechococcus sp. CCY9618 | Synechococcus elongatus PCC 7942 | Synechococcus elongatus UTEX 2973 | Synechococcus elongatus PCC 6301 | Synechococcus elongatus FACHB-242 | Synechococcus elongatus FACHB-1061 |
|---|---|---|---|---|---|---|---|---|---|---|
| J | 163 (4.7) | 161 (4.7) | 161 (4.7) | 161 (4.7) | 160 (5.1) | 163 (6.1) | 166 (6.1) | 165 (6.5) | 167 (6) | 167 (6) |
| K | 161 (4.8) | 155 (4.5) | 154 (4.5) | 138 (4.1) | 116 (3.7) | 114 (4.3) | 115 (4.2) | 110 (4.4) | 114 (4.1) | 114 (4.1) |
| L | 116 (3.4) | 121 (3.5) | 122 (3.5) | 127 (3.7) | 146 (4.7) | 108 (4.1) | 116 (4.3) | 112 (4.4) | 114 (4.1) | 114 (4.1) |
| B | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) | 2 (0.1) |
| D | 35 (1) | 42 (1.2) | 42 (1.2) | 37 (1.1) | 28 (0.9) | 27 (1) | 27 (1) | 25 (1) | 27 (1) | 27 (1) |
| V | 41 (1.2) | 43 (1.2) | 43 (1.2) | 51 (1.5) | 47 (1.5) | 31 (1.2) | 31 (1.1) | 31 (1.2) | 31 (1.1) | 31 (1.1) |
| T | 92 (2.7) | 100 (2.9) | 98 (2.8) | 89 (2.6) | 62 (2) | 85 (3.2) | 117 (4.3) | 109 (4.3) | 117 (4.2) | 117 (4.2) |
| M | 199 (5.9) | 201 (5.8) | 203 (5.9) | 204 (6) | 186 (6) | 134 (5) | 148 (5.4) | 145 (5.7) | 148 (5.3) | 148 (5.3) |
| N | 19 (0.6) | 21 (0.6) | 20 (0.6) | 17 (0.5) | 18 (0.6) | 26 (1) | 26 (1) | 26 (1) | 26 (0.9) | 25 (0.9) |
| U | 71 (2.1) | 73 (2.1) | 75 (2.2) | 71 (2.1) | 62 (2) | 34 (1.3) | 55 (2) | 53 (2.1) | 55 (2) | 54 (2) |
| O | 111 (3.3) | 114 (3.3) | 115 (3.3) | 114 (3.3) | 108 (3.5) | 94 (3.5) | 99 (3.6) | 101 (4) | 100 (3.6) | 100 (3.6) |
| C | 220 (6.5) | 229 (6.7) | 230 (6.7) | 220 (6.5) | 224 (7.2) | 199 (7.5) | 199 (7.3) | 202 (8) | 198 (7.2) | 199 (7.2) |
| G | 112 (3.3) | 111 (3.2) | 113 (3.3) | 104 (3.1) | 103 (3.3) | 71 (2.7) | 82 (3) | 80 (3.2) | 82 (3) | 82 (3) |
| E | 181 (5.4) | 184 (5.3) | 181 (5.3) | 174 (5.1) | 163 (5.2) | 138 (5.2) | 137 (5) | 132 (5.2) | 138 (5) | 138 (5) |
| F | 86 (2.6) | 89 (2.6) | 89 (2.6) | 88 (2.6) | 83 (2.7) | 98 (3.7) | 97 (3.6) | 96 (3.8) | 98 (3.5) | 98 (3.5) |
| H | 186 (5.5) | 183 (5.3) | 185 (5.4) | 185 (5.4) | 181 (5.8) | 167 (6.3) | 171 (6.3) | 171 (6.8) | 173 (6.3) | 173 (6.3) |
| I | 79 (2.3) | 80 (2.3) | 79 (2.3) | 88 (2.6) | 70 (2.3) | 54 (2) | 54 (2) | 51 (2) | 54 (2) | 54 (2) |
| P | 142 (4.2) | 151 (4.4) | 151 (4.4) | 162 (4.8) | 122 (3.9) | 152 (5.7) | 163 (6) | 157 (6.2) | 163 (5.9) | 164 (5.9) |
| Q | 46 (1.4) | 45 (1.3) | 45 (1.3) | 46 (1.4) | 39 (1.3) | 27 (1) | 44 (1.6) | 42 (1.7) | 44 (1.6) | 44 (1.6) |
| S | 669 (19.9) | 711 (20.7) | 710 (20.6) | 704 (20.7) | 629 (20.2) | 572 (21.5) | 737 (27.1) | 715 (28.3) | 745 (26.9) | 742 (36.8) |
Our sequenced sub-cluster 5.2 strains encode significantly more genes involved in carbohydrate (G), amino acid (E), and lipid (I) transport and metabolism than Synechococcus elongatus strains (p < .001, n = 5). Conversely, Synechococcus elongatus strains encode significantly more nucleotide transport and metabolism genes (F; p < .001, n = 5). As the Synechococcus elongatus genome size is smaller than that of our sub-cluster 5.2 freshwater strains, it may be expected to encode a reduced number of nucleotide-associated genes though this is not found. These genomic differences may be caused by the different environmental niches these two clades inhabit. Fresh waters are spatially diverse and exhibit a greater amount of nutrient heterogeneity than ocean environments [55]. Multiple other factors contribute to freshwater habitat niches, including light availability, temperature, water retention time, and composition of the surrounding microbial community [56]. However, while sub-cluster 5.2 and Synechococcus elongatus strains have been isolated from geographically distant locations, they occupy the same position in the water column (limnetic zone based on presence of phycocyanin [13]) and are more dominant in temperate waters. Increased genomic sequencing of taxa from sub-cluster 5.2 will aid in understanding freshwater picocyanobacteria ecology and the evolutionary context of these divergent lineages.
Further differences have been identified in the number of genes responsible for information storage and cellular processes between our sequenced strains and Synechococcus elongatus strains. Genes encoding defence mechanisms (V) and cell wall biogenesis-related (M) proteins are significantly increased in our newly sequenced strains (p < .05, n = 5). Meanwhile, Synechococcus elongatus strains have significantly higher numbers of genes involved in translation (J) and cell motility (N) (p < .001, n = 5). Research on cyanobacterial chemo- and photo-taxis has focused on Synechocystis spp. which exhibit a 'gliding' form of motility utilising a type IV pilus system [57]. Motility among marine Synechococcus spp. is achieved through multiple mechanisms, the most common through S-layer rotation [58,59], while recent findings have identified phototactic behaviour in Synechococcus elongatus [60]. However, the motility of sub-cluster 5.2 is yet to be determined. These differences in core cellular control may represent subtle changes in clade behaviour. As Synechococcus elongatus PCC 7942 is traditionally used as a model for freshwater Synechococcus, the variations in the genome may distort expectations of the Syn/Pro clade.
A comparison of the photosynthesis pathway between the newly sequenced picocyanobacteria and Synechococcus elongatus reveals a number of differences. Among core Photosystem II (PSII) components, the gene for the D2 protein (psbD) is surprisingly absent from the newly sequenced strains (in addition to two recently sequenced Synechococcus elongatus) (Table 3). The D2 protein forms part of the PSII reaction core alongside D1 (encoded by psbA) and is essential in binding the necessary redox-active cofactors for electron transfer [61]. The presence of psbD in other sub-cluster 5.2 strains is likewise unclear - absent from Synechococcus sp. BO8801 yet found in Synechococcus sp. 1G10 and Cyanobium gracile PCC 6307 (data not shown). However, the absence of psbD from our sequenced picocyanobacteria may be a result of the unclosed nature of the genome. psbC is found clustered with psbD in other cyanobacteria (e.g., Synechococcus elongatus PCC 7942 and Synechocystis sp. PCC 6803), though the contig encoding psbC in our sequenced Synechococcus spp. is truncated upstream (where the psbD locus is usually found). Other genes encoding photosynthesis electron transport proteins that are absent from our newly sequenced sub-cluster 5.2 strains include petL encoding the cytochrome b6f complex subunit 6, and petE encoding plastocyanin, responsible for transferring electrons from cytochrome b6f to Photosystem I (PSI). Cytochrome b6f is an intermediate in the transport of electrons from PSII to PSI, however the role of PetL in the complex is unclear. A function linked to stability of the dimeric state of the cytochrome b6f complex has been suggested while the non-essential nature of PetL in cyanobacteria has been demonstrated [62,63]. Accepting electrons from cytochrome b6f, copper-containing plastocyanin is another essential component of the photosynthesis electron transport chain. However, most cyanobacteria also contain Fe-containing cytochrome c6 (encoded by petJ). Expression of these two electron carriers is regulated by copper availability, a response to Fe-limitation [64]. The absence of plastocyanin in sub-cluster 5.2 strains appears to reduce adaptability in low-Fe environments, though heterocyst-forming cyanobacteria have been shown to preferentially utilise cytochrome c6 for electron transport, even in the presence of copper [65]. While the deletion of psbD is likely an artefact and must be resolved by the generation of closed freshwater picocyanobacteria genomes, further research to investigate the impact of the putative petL and petE gene deletions is necessary to elucidate this key physiological process in freshwater picocyanobacteria.
Genes encoding photosynthesis machinery and antennae proteins found in the five sequenced sub-cluster 5.2 Synechococcus genomes and five selected Synechococcus elongatus strains. Genes were identified through KEGG annotation. Copy number is indicated by the number of '+' symbols. Absence of the gene indicated by '-'.
| Synechococcus sp. | Synechococcus elongatus | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Kegg Orthology (KO) | Gene Product | CCAP 1479/9 | CCAP 1479/10 | CCAP 1479/13 | CCY 0621 | CCY 9618 | PCC 7942 | UTEX 2973 | PCC 6301 | FACHB-242 | FACHB-1061 |
| Photosynthesis | |||||||||||
| PSII | |||||||||||
| K02703 | PsbA | ++ | +++ | +++ | +++ | ++ | +++ | +++ | +++ | +++ | ++ |
| K02706 | PsbD | - | - | - | - | - | ++ | ++ | ++ | - | - |
| K02705 | PsbC | + | + | + | + | + | + | + | + | + | + |
| K02704 | PsbB | + | + | + | + | + | + | + | + | + | + |
| K02707 | PsbE | + | + | + | + | + | + | + | + | + | + |
| K02708 | PsbF | + | + | + | + | + | + | + | + | + | + |
| K02713 | PsbL | + | + | + | + | + | + | + | + | + | + |
| K02711 | PsbJ | + | + | + | + | + | + | + | + | + | + |
| K02712 | PsbK | + | + | + | + | + | + | + | + | + | + |
| K02714 | PsbM | + | + | + | + | + | + | + | + | + | + |
| K02709 | PsbH | + | + | + | + | + | + | + | + | + | + |
| K02710 | PsbI | + | + | + | + | + | + | - | + | - | - |
| K02716 | PsbO | + | + | + | + | + | + | + | + | + | + |
| K02717 | PsbP | + | + | + | + | + | + | + | + | + | + |
| K08901 | PsbQ | - | - | - | - | - | - | - | - | - | - |
| K03541 | PsbR | - | - | - | - | - | - | - | - | - | - |
| K03542 | PsbS | - | - | - | - | - | - | - | - | - | - |
| K02718 | PsbT | + | + | + | + | + | + | - | + | + | + |
| K02719 | PsbU | + | + | + | + | + | + | + | + | + | + |
| K02720 | PsbV | + | + | + | + | + | + | + | + | + | + |
| K02721 | PsbW | - | - | - | - | - | - | - | - | - | - |
| K02722 | PsbX | + | + | + | + | + | + | + | + | + | + |
| K02723 | PsbY | + | + | + | + | + | + | + | + | + | + |
| K02724 | PsbZ | + | + | + | + | ++ | + | + | + | + | + |
| K08902 | Psb27 | + | + | + | + | + | + | + | + | + | + |
| K08903 | Psb28 | + | + | + | + | + | + | + | + | + | + |
| K08904 | Psb28-2 | - | - | - | - | - | + | + | + | + | + |
| PSI | |||||||||||
| K02689 | PsaA | + | + | + | + | + | + | + | + | + | + |
| K02690 | PsaB | + | + | + | + | + | + | + | + | + | + |
| K02691 | PsaC | + | + | + | + | + | + | + | + | + | + |
| K02692 | PsaD | + | + | + | + | + | + | + | + | + | + |
| K02693 | PsaE | + | + | + | + | + | + | + | + | + | + |
| K02694 | PsaF | + | + | + | + | + | + | + | + | + | + |
| K08905 | PsaG | - | - | - | - | - | - | - | - | - | - |
| K02695 | PsaH | - | - | - | - | - | - | - | - | - | - |
| K02696 | PsaI | ++ | ++ | ++ | ++ | ++ | + | + | + | + | + |
| K02697 | PsaJ | + | + | + | + | + | + | + | + | + | + |
| K02698 | PsaK | + | + | + | + | + | ++ | ++ | ++ | ++ | ++ |
| K02699 | PsaL | + | + | + | + | + | + | + | + | + | + |
| K02700 | PsaM | + | + | + | + | + | + | + | + | + | + |
| K02701 | PsaN | - | - | - | - | - | - | - | - | - | - |
| K14332 | PsaO | - | - | - | - | - | - | - | - | - | - |
| K02702 | PsaX | - | - | - | - | - | - | - | - | - | - |
| Cytochrome b6/f complex | |||||||||||
| K02635 | PetB | + | + | + | + | + | + | + | + | + | + |
| K02637 | PetD | + | + | + | + | + | + | + | + | + | + |
| K02634 | PetA | + | + | + | + | + | + | + | + | + | + |
| K02636 | PetC | + | + | + | ++ | + | + | + | + | + | + |
| K02642 | PetL | - | - | - | - | - | + | + | + | + | + |
| K02643 | PetM | + | + | + | + | + | + | + | + | + | + |
| K03689 | PetN | + | + | + | + | + | + | - | + | + | + |
| K02640 | PetG | + | + | + | + | + | + | + | - | + | + |
| Photosynthetic electron transport | |||||||||||
| K02638 | PetE | - | - | - | - | - | + | + | + | + | + |
| K02639 | PetF | ++++ | ++++ | ++++ | ++++ | ++++ | +++ | +++ | +++ | +++ | +++ |
| K02641 | PetH | + | + | + | + | + | + | + | + | + | + |
| K08906 | PetJ | + | + | + | ++ | ++ | +++ | +++ | +++ | +++ | +++ |
| F-type ATPase | |||||||||||
| K02112 | beta | + | + | + | + | + | + | + | + | + | + |
| K02111 | alpha | + | + | + | + | + | + | + | + | + | + |
| K02115 | gamma | + | + | + | + | + | + | + | + | + | + |
| K02113 | delta | + | + | + | + | + | + | + | + | + | + |
| K02114 | epsilon | + | + | + | + | + | + | + | + | + | + |
| K02110 | c | + | + | + | + | + | + | + | + | + | + |
| K02108 | a | + | + | + | + | + | + | + | + | + | + |
| K02109 | b | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ |
| Photosynthesis - Antenna Proteins | |||||||||||
| Allophycocyanin (AP) | |||||||||||
| K02092 | ApcA | + | + | + | + | + | + | + | + | + | + |
| K02093 | ApcB | + | + | + | + | + | + | + | + | + | + |
| K02094 | ApcC | + | + | + | + | + | + | + | + | + | + |
| K02095 | ApcD | + | + | + | + | + | ++ | ++ | + | ++ | ++ |
| K02096 | ApcE | + | + | + | + | + | + | + | + | + | + |
| K02097 | ApcF | + | + | + | + | + | + | + | + | + | + |
| Phycocyanin (PC)/Phycoerythrocyanin (PEC) | |||||||||||
| K02284 | CpcA | ++ | ++ | ++ | + | - | ++ | ++ | ++ | ++ | ++ |
| K02285 | CpcB | +++ | +++ | +++ | ++ | + | ++ | ++ | ++ | ++ | ++ |
| K02286 | CpcC | - | - | - | - | - | ++ | ++ | ++ | ++ | ++ |
| K02287 | CpcD | + | + | + | + | + | + | + | + | + | + |
| K02288 | CpcE | + | + | + | + | + | + | + | + | + | + |
| K02289 | CpcF | + | + | + | + | + | + | + | + | + | + |
| K02290 | CpcG | ++ | ++ | ++ | ++ | ++ | + | + | + | + | + |
| Phycoerythrin (PE) | |||||||||||
| K05376 | CpeA | - | - | - | - | - | - | - | - | - | - |
| K05377 | CpeB | - | - | - | - | - | - | - | - | - | - |
| K05378 | CpeC | ++ | ++ | ++ | ++ | ++ | - | - | - | - | - |
| K05379 | CpeD | - | - | - | - | - | - | - | - | - | - |
| K05380 | CpeE | - | - | - | - | - | - | - | - | - | - |
| K05381 | CpeR | - | - | - | - | - | - | - | - | - | - |
| K05382 | CpeS | - | - | - | - | - | + | + | + | + | + |
| K05383 | CpeT | - | - | - | - | - | - | - | - | - | - |
| K05384 | CpeU | - | - | - | - | - | - | - | - | - | - |
| K05385 | CpeY | - | - | - | - | - | - | - | - | - | - |
| K05386 | CpeZ | - | - | - | - | - | - | - | - | - | - |
In addition to core photosynthetic electron transport apparatus, the copy number and composition of antennae proteins comprising the light-harvesting phycobilisome (PBS) displays subtle differences (Table 3). Synechococcus elongatus strains encode two copies of apcD, encoding a key component of the allophycocyanin (AP) central core of PBS, though our newly sequenced sub-cluster 5.2 picocyanobacteria encode solely apcD1. The role of ApcD has been shown to slightly vary between Synechococcus elongatus PCC 7942 and another cyanobacterial model organism - Synechocystis sp. PCC 6803. ApcD is vital for efficient energy transfer from the PBS to PSI in Synechococcus elongatus PCC 7942 while the lack of ApcD has no impact on PSI energy transfer in Synechocystis sp. PCC 6803, instead inhibiting state transitions in response to unbalanced light conditions [66]. Furthermore, multiple copies of apcD have been linked to photoacclimation to far-red light (700 - 750 nm), aiding absorbance of a greater diversity of wavelengths [67]. This may suggest a wider range of utilisable wavelengths for Synechococcus elongatus strains, resulting in community shifts in heavily shaded areas.
There are more significant variations in the encoding of phycobiliprotein-rods which radiate out from the PBS core. There are differences in the copy number of phycocyanin (PC) subunits cpcA and cpcB with Synechococcus sp. CCY9618 encoding only cpcB. Other newly sequenced genomes encode both subunits with cpcB at an increased copy number compared to Synechococcus elongatus strains (Table 3). Interestingly, cpcC is absent from our sub-cluster 5.2 strains. This encodes the LR33 PC-associated linker polypeptide, responsible for stabilising rod substructures [68]. Meanwhile, the same strains encode an additional copy of cpcG (encoding a linker protein required for rod attachment to the AP core), with the two copies having distinct roles in PSII (cpcG1) and PSI (cpcG2) in Synechocystis sp. PCC 6803[69]. The absence of cpcG2 in Synechococcus elongatus strains suggests further differences in photosynthetic machinery between the two groups. Furthermore, while phycoerythrin (PE) is known to be absent in Synechococcus elongatus strains, it has been observed in other sequenced sub-cluster 5.2 freshwater picocyanobacteria [13]. However, the strains sequenced in this study are absent of cpeAB indicating PBS rods of PC only. Though lacking PE subunits, freshwater Synechococcus encode various PE-associated proteins. Our sequenced Synechococcus encode two copies of cpeC, a PE-associated rod linker protein, while Synechococcus elongatus encode cpeS, an S-type lyase essential for mature PE generation [70,71]. It is unclear if these genes are expressed, and the function they provide for Synechococcus lacking PE.
The most abundant N source in fresh water is nitrate [72], a nutrient which cyanobacteria can access via the narB-nrtABCD-nirA operon. This operon encodes the necessary proteins for nitrate assimilation, yet the gene neighbourhood of this operon differs between sub-cluster 5.2 freshwater picocyanobacteria and Synechococcus elongatus. This operon consists of a nitrate/nitrite bi-specific ABC-type transporter (nrtABCD), nitrate reductase (narB), and nitrite reductase (nirA). Among our newly sequenced strains (apart from Synechococcus sp. CCY 9618), nirA and narB are transcribed in the opposite direction to nrtABCD whereas Synechococcus elongatus encodes the six core genes continuously (Figure 3). Furthermore, there are unrelated genes flanking nrtABCD - anthranilate phosphoribosyltransferase and a hypothetical gene. Contiguous operons are known for rapid gene expression for all proteins of a specific cellular process, however the unassociated genes and two-way transcription may suggest sub-cluster 5.2 freshwater picocyanobacteria respond slower to nitrate inducement, though bidirectional promoters may be involved.
Gene neighbourhood of the narB-nrtABCD-nirA operon for nitrate assimilation. Green arrows are genes involved with nitrate assimilation. Orange arrows are genes involved with photosynthesis. Blue arrows are genes involved with carbon metabolism. Black arrows are other annotated genes while grey arrows indicate hypothetical genes. petA: apocytochrome f (K02634). petC: cytochrome b6f complex iron-sulphur subunit (K02636), psaJ: photosystem I subunit 9 (K02697), psaF: Photosystem I subunit 3 K02694), xfp: xylulose-5-phosphate/fructose-6-phosphate phosphoketolase (K01621), ackA: acetate kinase (K00925), sdhB: succinate dehydrogenase/fumarate reductase iron-sulphur subunit (K00240), sdhA: succinate dehydrogenase/fumarate reductase flavoprotein subunit (K00239), sdhC: succinate dehydrogenase/fumarate reductase cytochrome b subunit (K00241).
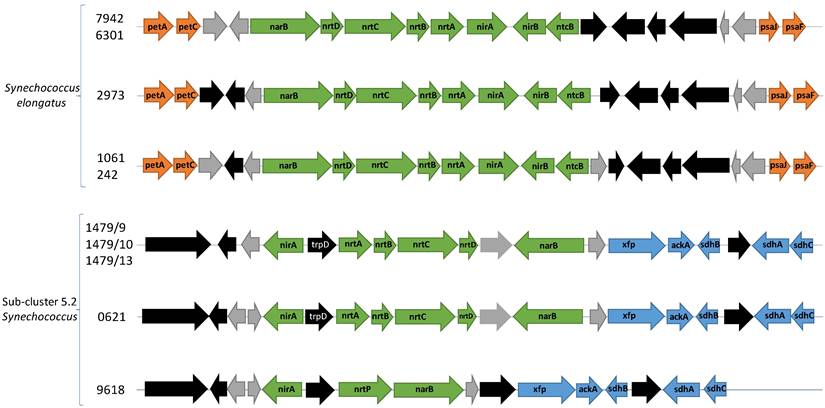
Additional genes involved with nitrite assimilation are found in Synechococcus elongatus strains but absent from our sequenced strains. These include nirB, required for maximal nitrite reductase activity, and ntcB, a transcription factor involved in nitrite-induced gene activation [73,74]. Though nitrate is the most abundant traditional N source, it is also the most energetically costly, requiring eight electrons to reduce fully to ammonium (nitrate > nitrite > ammonium). Increasing the preference for nitrite over nitrate can reduce this demand which may result in substantial energy savings. Synechococcus sp. CCY9618 encodes a homologous transporter previously only identified in marine picocyanobacteria, nrtP, which preferentially takes up nitrate over nitrite [75]. The differences between sub-cluster 5.2 freshwater picocyanobacteria and Synechococcus elongatus may indicate differing preferences for nutrient growth, influencing the composition of the Synechococcus community.
The newly sequenced five freshwater picocyanobacteria expand the number of genomes available for sub-cluster 5.2 of the Syn/Pro clade. The number of genomic capabilities for metabolism and cellular processes vary significantly between these strains and Synechococcus elongatus strains. These findings contribute to a better understanding not only of the ecology, but the evolutionary relationships of freshwater Synechococcus and re-evaluates the conclusions that can be drawn from the model organism Synechococcus elongatus.
Abbreviations
AP: allophycocyanin; CCGs: core cyanobacterial genes; PBS: phycobilisome; PE: phycoerythrin; PSI: Photosystem I; PSII: Photosystem II.
Supplementary Material
Supplementary tables.
Supplementary figures 1, 3-7.
Supplementary figure 2.
Acknowledgements
Funding support for this work came from a NERC CDT scholarship (NE/RO11524/1) for E. J. Druce and a Royal Society University Research Fellowship to P. S.-B.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Steitz A, Velimirov B. Contribution of Picocyanobacteria to total primary production and community respiratory losses in a backwater system. J Plankton Res. 1999;21:2341-60
2. Schmidt K, Birchill AJ, Atkinson A. et al. Increasing picocyanobacteria success in shelf waters contributes to long-term food web degradation. Glob Chang Biol. 2020;26:5574-87
3. Romero-Viana L, Keely BJ, Camacho A, Vicente E, Miracle MR. Primary production in Lake La Cruz (Spain) over the last four centuries: Reconstruction based on sedimentary signal of photosynthetic pigments. J Paleolimnol. 2010;43:771-86
4. Waleron M, Waleron K, Vincent WF, Wilmotte A. Allochthonous inputs of riverine picocyanobacteria to coastal waters in the Arctic Ocean. FEMS Microbiol Ecol. 2007;59:356-65
5. Flombaum P, Gallegos JL, Gordillo RA. et al. Present and future global distributions of the marine Cyanobacteria Prochlorococcus and Synechococcus. Proc Natl Acad Sci U S A. 2013;110:9824-9
6. Huang S, Liu Y, Hu A. et al. Genetic diversity of picocyanobacteria in Tibetan lakes: Assessing the endemic and universal distributions. Appl Environ Microbiol. 2014;80:7640-50
7. Stockner J, Callieri C, Cronberg G. Picoplankton and Other Non-Bloom-Forming Cyanobacteria in Lakes. Ecol Cyanobacteria. 2000:195-231
8. Vörös L, Callieri C, V-Balogh K, Bertoni R. Freshwater picocyanobacteria along a trophic gradient and light quality range. Hydrobiologia. 1998;369-370:117-25
9. Sánchez-Baracaldo P, Handley BA, Hayest PK. Picocyanobacterial community structure of freshwater lakes and the Baltic Sea revealed by phylogenetic analyses and clade-specific quantitative PCR. Microbiology. 2008;154:3347-57
10. Becker S, Sánchez-Baracaldo P, Singh AK, Hayes PK. Spatio-temporal niche partitioning of closely related picocyanobacteria clades and phycocyanin pigment types in Lake Constance (Germany). FEMS Microbiol Ecol. 2012;80:488-500
11. Cabello-Yeves PJ, Picazo A, Camacho A. et al. Ecological and genomic features of two widespread freshwater picocyanobacteria. Environ Microbiol. 2018;20:3757-71
12. Di Cesare A, Cabello-Yeves PJ, Chrismas NAM, Sánchez-Baracaldo P, Salcher MM, Callieri C. Genome analysis of the freshwater planktonic Vulcanococcus limneticus sp. nov. reveals horizontal transfer of nitrogenase operon and alternative pathways of nitrogen utilization. BMC Genomics. 2018;19:1-12
13. Sánchez-Baracaldo P, Bianchini G, Di Cesare A, Callieri C, Chrismas NAM. Insights Into the Evolution of Picocyanobacteria and Phycoerythrin Genes (mpeBA and cpeBA). Front Microbiol. 2019;10:1-17
14. Scanlan DJ, Ostrowski M, Mazard S. et al. Ecological Genomics of Marine Picocyanobacteria. Microbiol Mol Biol Rev. 2009;73:249-99
15. Ahlgren NA, Belisle BS, Lee MD. Genomic mosaicism underlies the adaptation of marine Synechococcus ecotypes to distinct oceanic iron niches. Environ Microbiol. 2020;22:1801-15
16. Yelton AP, Acinas SG, Sunagawa S, Bork P, Pedrós-Alió C, Chisholm SW. Global genetic capacity for mixotrophy in marine picocyanobacteria. ISME J. 2016;10:2946-57
17. Glibert PM, Heil CA, Madden CJ, Kelly SP. Dissolved organic nutrients at the interface of fresh and marine waters: flow regime changes, biogeochemical cascades and picocyanobacterial blooms—the example of Florida Bay, USA. Biogeochemistry [Internet]. 2021 4. Available at: https://doi.org/10.1007/s10533-021-00760-4
18. Cabello-Yeves PJ, Callieri C, Picazo A. et al. Elucidating the picocyanobacteria salinity divide through ecogenomics of new freshwater isolates. BMC Biol [Internet]. 2022;20:1-24 Available at: https://doi.org/10.1186/s12915-022-01379-z
19. Jaiswal D, Sengupta A, Sengupta S, Madhu S, Pakrasi HB, Wangikar PP. A Novel Cyanobacterium Synechococcus elongatus PCC 11802 has Distinct Genomic and Metabolomic Characteristics Compared to its Neighbor PCC 11801. Sci Rep. 2020;10:1-15
20. Gorelova OA, Baulina OI, Rasmussen U, Koksharova OA. The pleiotropic effects of ftn2 and ftn6 mutations in cyanobacterium Synechococcus sp. PCC 7942: An ultrastructural study. Protoplasma. 2013;250:931-42
21. Vázquez-Bermüdez MF, Paz-Yepes J, Herrero A, Flores E. The NtcA-activated amt1 gene encodes a permease required for uptake of low concentrations of ammonium in the cyanobacterium Synechococcus sp. PCC 7942. Microbiology. 2002;148:861-9
22. Paz-Yepes J, Herrero A, Flores E. The NtcA-regulated amtB gene is necessary for full methylammonium uptake activity in the cyanobacterium Synechococcus elongatus. J Bacteriol. 2007;189:7791-8
23. Omata T, Andriesse X, Hirano A. Identification and characterization of a gene cluster involved in nitrate transport in the cyanobacterium Synechococcus sp. PCC7942. MGG Mol Gen Genet. 1993;236:193-202
24. Maeda S, Aoba R, Nishino Y, Omata T. A Novel Bacterial Nitrate Transporter Composed of Small Transmembrane Proteins. Plant Cell Physiol. 2019;0:1-13
25. Escudero L, Mariscal V, Flores E. Functional dependence between septal protein SepJ from Anabaena sp. Strain PCC 7120 and an Amino Acid ABC-Type Uptake Transporter. J Bacteriol. 2015;197:2721-30
26. Robertson BR, Tezuka N, Watanabe MM. Phylogenetic analyses of Synechococcus strains (cyanobacteria) using sequences of 16S rDNA and part of the phycocyanin operon reveal multiple evolutionary lines and reflect phycobilin content. Int J Syst Evol Microbiol. 2001;51:861-71
27. Callieri C, Coci M, Corno G. et al. Phylogenetic diversity of nonmarine picocyanobacteria. FEMS Microbiol Ecol. 2013;85:293-301
28. Stanier RY, Kunisawa R, Mandel M, Cohen-Bazire G. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol Rev. 1971;35:171-205
29. Bolger AM, Lohse M, Usadel B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30:2114-20
30. Bankevich A, Nurk S, Antipov D. et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19:455-77
31. Mulkidjanian AY, Koonin E V, Makarova KS. et al. The cyanobacterial genome core and the origin of photosynthesis. Proc Natl Acad Sci U S A. 2006;103:13126-31
32. Chrismas NAM, Barker G, Anesio AM, Sánchez-Baracaldo P. Genomic mechanisms for cold tolerance and production of exopolysaccharides in the Arctic cyanobacterium Phormidesmis priestleyi BC1401. BMC Genomics [Internet]. 2016;17:1-14 Available at: http://dx.doi.org/10.1186/s12864-016-2846-4
33. Wick RR, Schultz MB, Zobel J, Holt KE. Bandage: Interactive visualization of de novo genome assemblies. Bioinformatics. 2015;31:3350-2
34. Besemer J, Borodovsky M. GeneMark: Web software for gene finding in prokaryotes, eukaryotes and viruses. Nucleic Acids Res. 2005;33:451-4
35. Hyatt D, Chen G-L, LoCascio PF, Land ML, Larimer FW, Hauser LJ. Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinformatics [Internet]. 2010;11:1-8 Available at: https://doi.org/10.1186/1471-2105-11-119
36. Nawrocki EP, Eddy SR. Infernal 1.1: 100-fold faster RNA homology searches. Bioinformatics. 2013;29:2933-5
37. Chan PP, Lowe TM. tRNAscan-SE: Searching for tRNA genes in genomic sequences. Methods Mol Biol. 2019;1962:1-14
38. Simão FA, Waterhouse RM, Ioannidis P, Kriventseva E V, Zdobnov EM. BUSCO: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics. 2015;31:3210-2
39. Chen IMA, Chu K, Palaniappan K. et al. IMG/M v.5.0: An integrated data management and comparative analysis system for microbial genomes and microbiomes. Nucleic Acids Res. 2019;47:D666-77
40. Huerta-Cepas J, Forslund K, Coelho LP. et al. Fast genome-wide functional annotation through orthology assignment by eggNOG-mapper. Mol Biol Evol. 2017;34:2115-22
41. Kanehisa M, Goto S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000;28:27-30
42. Sánchez-Baracaldo P. Origin of marine planktonic cyanobacteria. Sci Rep. 2015;5:14-7
43. Boden JS, Konhauser KO, Robbins LJ, Sánchez-Baracaldo P. Timing the evolution of antioxidant enzymes in cyanobacteria. Nat Commun [Internet]. 2021 12. Available at: http://dx.doi.org/10.1038/s41467-021-24396-y
44. Blank CE, Sanchez-Baracaldo P. Timing of morphological and ecological innovations in the cyanobacteria - A key to understanding the rise in atmospheric oxygen. Geobiology. 2010;8:1-23
45. Camacho C, Coulouris G, Avagyan V. et al. BLAST+: Architecture and applications. BMC Bioinformatics. 2009;10:1-9
46. Katoh K, Standley DM. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol [Internet]. 2013;30:772-80 Available at: https://doi.org/10.1093/molbev/mst010
47. Minh BQ, Schmidt HA, Chernomor O. et al. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol Biol Evol. 2020;37:1530-4
48. Kalyaanamoorthy S, Minh BQ, Wong TKF, Von Haeseler A, Jermiin LS. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat Methods. 2017;14:587-9
49. Chernomor O, Von Haeseler A, Minh BQ. Terrace Aware Data Structure for Phylogenomic Inference from Supermatrices. Syst Biol. 2016;65:997-1008
50. Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Molecular biology and evolution. Mol Biol Evol. 2017;35:518-22
51. Giovannoni SJ, Cameron Thrash J, Temperton B. Implications of streamlining theory for microbial ecology. ISME J. 2014;8:1553-65
52. Mann S, Chen YPP. Bacterial genomic G + C composition-eliciting environmental adaptation. Genomics [Internet]. 2010;95:7-15 Available at: http://dx.doi.org/10.1016/j.ygeno.2009.09.002
53. Weissman JL, Fagan WF, Johnson PLF. Linking high GC content to the repair of double strand breaks in prokaryotic genomes. PLoS Genet [Internet]. 2019;15:1-19 Available at: http://dx.doi.org/10.1371/journal.pgen.1008493
54. Berube PM, Rasmussen A, Braakman R, Stepanauskas R, Chisholm SW. Emergence of trait variability through the lens of nitrogen assimilation in Prochlorococcus. Elife. 2019;8:1-28
55. Elser JJ, Bracken MES, Cleland EE. et al. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett. 2007;10:1135-42
56. Callieri C. Single cells and microcolonies of freshwater picocyanobacteria: A common ecology. J Limnol. 2010;69:257-77
57. Menon SN, Varuni P, Bunbury F, Bhaya D, Menon GI. Phototaxis in Cyanobacteria: From Mutants to Models of Collective Behavior. MBio. 2021 12
58. McCarren J, Brahamsha B. Swimming motility mutants of marine Synechococcus affected in production and localization of the S-layer protein SwmA. J Bacteriol. 2009;191:1111-4
59. Callieri C, Cabello-Yeves PJ, Bertoni F. The “Dark Side” of Picocyanobacteria: Life as We Do Not Know It (Yet). Microorganisms. 2022;10:1-18
60. Yang Y, Lam V, Adomako M. et al. Phototaxis in a wild isolate of the cyanobacterium Synechococcus elongatus. Proc Natl Acad Sci U S A. 2018;115:E12378-87
61. Kiss É, Kós PB, Chen M, Vass I. A unique regulation of the expression of the psbA, psbD, and psbE genes, encoding the D1, D2 and cytochrome b559 subunits of the Photosystem II complex in the chlorophyll d containing cyanobacterium Acaryochloris marina. Biochim Biophys Acta - Bioenerg [Internet]. 2012;1817:1083-94 Available at: http://dx.doi.org/10.1016/j.bbabio.2012.04.010
62. Breyton C, Tribet C, Olive J, Dubacq JP, Popott JL. Dimer to monomer conversion of the cytochrome b6f complex: Causes and consequences. J Biol Chem. 1997;272:21892-900
63. Schneider D, Volkmer T, Rögner M. PetG and PetN, but not PetL, are essential subunits of the cytochrome b6f complex from Synechocystis PCC 6803. Res Microbiol. 2007;158:45-50
64. García-Cañas R, Giner-Lamia J, Florencio FJ, López-Maury L. A protease-mediated mechanism regulates the cytochrome c6/plastocyanin switch in Synechocystis sp. PCC 6803. Proc Natl Acad Sci U S A. 2021 118
65. Torrado A, Ramírez-Moncayo C, Navarro JA, Mariscal V, Molina-Heredia FP. Cytochrome c6 is the main respiratory and photosynthetic soluble electron donor in heterocysts of the cyanobacterium Anabaena sp. PCC 7120. Biochim Biophys Acta - Bioenerg [Internet]. 2019;1860:60-8 Available at: https://doi.org/10.1016/j.bbabio.2018.11.009
66. Calzadilla PI, Muzzopappa F, Sétif P, Kirilovsky D. Different roles for ApcD and ApcF in Synechococcus elongatus and Synechocystis sp. PCC 6803 phycobilisomes. Biochim Biophys Acta - Bioenerg [Internet]. 2019;1860:488-98 Available at: https://doi.org/10.1016/j.bbabio.2019.04.004
67. Xu QZ, Han JX, Tang QY. et al. Far-red light photoacclimation: Chromophorylation of FR induced α- and β-subunits of allophycocyanin from Chroococcidiopsis thermalis sp. PCC7203. Biochim Biophys Acta - Bioenerg [Internet]. 2016;1857:1607-16 Available at: http://dx.doi.org/10.1016/j.bbabio.2016.06.008
68. de Lorimier R, Bryant DA, Stevens SE. Genetic analysis of a 9 kDa phycocyanin-associated linker polypeptide. BBA - Bioenerg. 1990;1019:29-41
69. Kondo K, Xiao XG, Katayama M, Ikeuchi M. Distinct roles of CpcG1 and CpcG2 in phycobilisome assembly in the cyanobacterium Synechocystis sp. PCC 6803. Photosynth Res. 2005;84:269-73
70. Bezy RP, Wiltbank L, Kehoe DM. Light-dependent attenuation of phycoerythrin gene expression reveals convergent evolution of green light sensing in cyanobacteria. Proc Natl Acad Sci U S A. 2011;108:18542-7
71. Wiethaus J, Busch AWU, Kock K, Leichert LI, Herrmann C, Frankenberg-Dinkel N. CpeS is a lyase specific for attachment of 3Z-PEB to Cys82 of β-phycoerythrin from Prochlorococcus marinus MED4. J Biol Chem. 2010;285:37561-9
72. Follett RF, Hatfield JL. Nitrogen in the environment: sources, problems, and management. ScientificWorldJournal. 2001;1(Suppl 2):920-6
73. Frías JE, Flores E. Negative regulation of expression of the nitrate assimilation nirA operon in the heterocyst-forming cyanobacterium Anabaena sp. strain PCC 7120. J Bacteriol. 2010;192:2769-78
74. Aichi M, Takatani N, Omata T. Role of NtcB in activation of nitrate assimilation genes in the cyanobacterium Synechocystis sp. strain PCC 6803. J Bacteriol. 2001;183:5840-7
75. Aichi M, Yoshihara S, Yamashita M, Maeda SI, Nagai K, Omata T. Characterization of the nitrate-nitrite transporter of the major facilitator superfamily (the nrtP gene product) from the cyanobacterium Nostoc punctiforme strain ATCC 29133. Biosci Biotechnol Biochem. 2006;70:2682-9
Author contact
Corresponding author: Name: Patricia Sánchez-Baracaldo, E-mail: p.sanchez-baracaldoac.uk.

 Global reach, higher impact
Global reach, higher impact