ISSN: 1839-9940
J Genomics 2023; 11:14-19. doi:10.7150/jgen.80829 This volume Cite
Research Paper
Draft Genomes of Halophilic Chromohalobacter and Halomonas Strains Isolated from Brines of The Carpathian Foreland, Poland
1. Department of Molecular Microbiology, Faculty of Biology and Environmental Protection, University of Lodz, Lodz, Poland
2. Biobank Lab, Department of Oncobiology and Epigenetics, Faculty of Biology and Environmental Protection, University of Lodz. Lodz, Poland
3. Screening of Biological Activity Assays and Collection of Biological Material Laboratory, Wroclaw Medical University Biobank, Faculty of Pharmacy, Wroclaw Medical University, Wroclaw, Poland
Received 2022-11-14; Accepted 2023-2-13; Published 2023-3-21
Abstract
Chromohalobacter and Halomonas are genera of bacterial microorganisms belonging to the group of halophiles. They are characterized by high diversity and the ability to produce bioproducts of biotechnological importance, such as ectoine, biosurfactants and carotenoids. Here, we report three draft genomes of Chromohalobacter and two draft genomes of Halomonas isolated from brines. The length of the genomes ranged from 3.6 Mbp to 3.8 Mbp, and GC content was in the 60.11%-66.46% range. None of the analysed genomes has been assigned to any previously known species of the genus Chromohalobacter or Halomonas. Phylogenetic analysis revealed that Chromohalobacter 296-RDG and Chromohalobacter 48-RD10 belonged to the same species, and Chromohalobacter 11-W is more distantly related to the other two analysed strains than to Chromohalobacter canadensis. Halomonas strains 11-S5 and 25-S5 were clustered together and located close to Halomonas ventosae. Functional analysis revealed BGCs related to ectoine production in all genomes analysed. This study increases our overall understanding of halophilic bacteria and is also consistent with the notion that members of this group have significant potential as useful natural product producers.
Introduction
Hypersaline ecosystems have been widely explored mainly because of their unique biodiversity and biotechnological potential. It is related to the adaptation of halophiles to the conditions of high osmotic pressure, limited availability of energy resources and other unfavourable environmental circumstances (1). Novel halophilic microorganisms have been isolated in recent years from habitats such as saline lakes, salt mines, saline soils or fermented foods (2-6). A great variety of saline environments related to factors other than salinity itself, such as pH, temperature or the availability of nutrients, results in a considerable diversity of inhabiting microorganisms, both in terms of genetics and metabolism.
Halomonadaceae is a family of halophilic Gammaproteobacteria, including Chromohalobacter, Halomonas and 12 other genera (7,8). Currently, Chromohalobacter genus includes eight validly published species isolated from salterns, seas and food products. The process of validating the publication of new species of prokaryotes is related to the fulfilment of all of the requirements set out in the “International Code of Nomenclature of Prokaryotes” (9). Moreover, the new species Chromohalobacter moromii sp. nov. isolated from lupine-based moromi fermentation has been described and is pending validation (6). Microorganisms belonging to the genus Chromohalobacter, for example, Chromohalobacter salexigens, have been identified as producers of biotechnologically valuable compounds and, due to their genomic characteristics, could become a useful metabolic engineering tool for the overproduction of ectoines (10).
On the other hand, the Halomonas genus includes 117 validly published species and new species like Halomonas alkalisoli sp. nov are waiting for validation (2). Most of these species are widely distributed in saline habitats, such as salt lakes, marine environments, and saline soils (11,12). Halomonas strains exhibit high metabolic and physiological versatility, thus a wide range of bioproducts, such as ectoine, glycine betaine and polyhydroxyalkanoates (PHA) can be produced (12,13).
This study presents the characteristics of five genomes of microorganisms isolated from brines, sources of which are located in the southern part of the Carpathian Foreland in Poland near Kraków city. Three genome sequences of Chromohalobacter sp. and two draft genome sequences of Halomonas sp are reported.
Materials and methods
Three strains of Chromohalobacter sp. and two strains of Halomonas sp. were isolated from brines. Chromohalobacter 11-W was isolated from the borehole of the former Barycz mining area (49◦59'05” N 20◦00'52” E), Chromohalobacter 296-RDG and Chromohalobacter 48-RD10 were isolated from the Bochnia Salt Mine (49°58′09″N 20°25′03″E) and Halomonas strains 11-S5 and 25-S5 were isolated from the brine source in Łapczyca (49°57'30"N 20°21'41"E). Strains were cultured in 28°C on plates containing halobacteria medium (DSMZ 372) with 15% NaCl addition for strains Chromohalobacter 11-W, Chromohalobacter 296-RDG and Halomonas 25-S5. Rest of the strains were cultured in the same conditions except NaCl concentration which was changed to 20%. Medium was solidified with 2% agar. For genomic DNA extraction QIAamp DNA Mini Kit (Qiagen, Hilden, Germany) has been used.
Paired-end libraries were prepared from 1 ng of high-quality genomic DNA with the Nextera XT DNA sample preparation kit according to the manufacturer's instructions (Illumina Inc., San Diego, USA). The libraries were sequenced using a NextSeq 500 instrument (Illumina, San Diego, USA) at a read length of 2 × 150 bp in Biobank Lab, University of Lodz. The quality of reads was checked using FastQC (14). Furthermore, adaptors and low-quality sequences were removed from the reads with trim galore v. 0.6.4 on default parameters (15). De novo assembly was performed with SPAdes v3.15.0 (16). Contigs with coverage lower than 2, or lengths lower than 500 bp, were removed from the assembly. Contamination and completeness of assemblies were calculated using CheckM based on a reference database of marker genes (17). Overall statistics of assemblies quality parameters were tested using Quast (18). Genomes annotation was performed using Prokka v.1.14.0 (19) and the eggNOG-mapper website v.5.0.0 (20). Biosynthetic gene clusters (BGCs) prediction was conducted with antiSMASH v.6.0.1 (21). Taxonomic annotation of genomes was performed with gtdb-tk version 1.5.1 (22). Phylogenomic analysis was accomplished on Type (Strain) Genome Server (TYGS) (23), and the whole-genome sequence-based phylogenetic tree was visualized using iTOL (24). For TYGS analysis, 27 reference strain genomes were used. They are available in the NCBI database under accessions: NC_007963, NZ_BNAE01000000, NZ_CAAHFN010000000, NZ_CABVOU010000000, NZ_CP018139, NZ_FNIV01000000, NZ_FOBC01000000, NZ_FPAQ01000000, NZ_JACHXQ010000000, NZ_JACHXR010000000, NZ_JACHZF010000000, NZ_JAGXFD010000000, NZ_JAKGAJ010000000, NZ_JAKGAK010000000, NZ_JAKGAL010000000, NZ_JAKGAM010000000, NZ_JAKGAN010000000, NZ_PYVX01000000, NZ_PZJV01000000, NZ_QPIJ01000000, NZ_RXNS01000000, NZ_SDMO01000000, NZ_SNZJ01000000, NZ_SOBR01000000, NZ_VBUI01000000, NZ_WUTS01000000, NZ_WUTT01000000. Phylogenetic tree for 16S rRNA gene sequences was prepared in MEGA X software (25).
All sequencing data are publicly available from the National Institutes of Health under BioProject accessions PRJNA899688, PRJNA899690, PRJNA899692, PRJNA899693, PRJNA899694.
Results and discussion
Each draft genome was composed of between 38 and 321 contigs, with genome sizes ranging 3.6-3.9 Mbps. The overall genome completeness was estimated at between 98.71-99.86%, with contamination in the range of 0.54-8.42 and GC content in the 60.11-66.46% range. The summary is presented in Table 1.
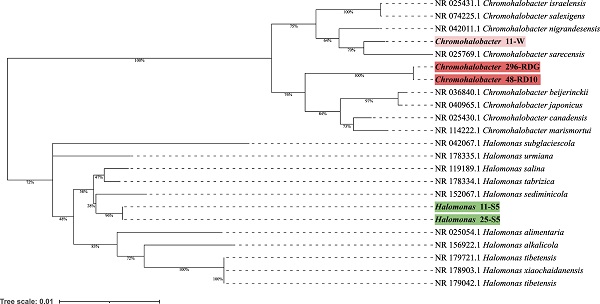
Preliminary taxonomic annotation of genomes using gtdb-tk, assigned isolates 11-W, 296-RDG, and 48-RD10 to Chromohalobacter genus, and isolates 11-S5, 25-S5 to Halomonas genus. The assignment to the species level was impossible because of too high differences in genome sequences between the analysed strains and the previously described genomes available in the databases. A phylogenetic analysis was performed in order to deepen knowledge about the relationship between the analysed isolates and other species. A phylogenetic tree, based on the 16S rRNA gene sequences was built. The resulting tree confirmed that the isolates 11-W, 296-RDG, and 48-RD10 fell within a cluster comprising members of the genus Chromohalobacter and the strains 11-S5, 25-S5 fell within a cluster including members of the genus Halomonas (Figure 1). However, in both cases, the analysed strains were separated from the other species included in the analysis. The closest species for Chromohalobacter strains 296-RDG and 48-RD10 was Chromohalobacter canadensis, and for strain 11-W, it was Chromohalobacter sarecensis. In the case of Halomonas strains, the closest taxon was Halomonas sediminicola. This initial phylogenetic analysis, using a comparison of 16S rRNA gene sequences, was then deepened through the construction of a further, whole-genome sequence-based phylogenetic tree build using Genome BLAST Distance Phylogeny approach (GBDP) created on the TYGS platform (Figure 2). The obtained phylogenetic tree confirmed the observations made at an earlier stage. On the genome-wide scale, it was noticed that Chromohalobacter 11-W is more distant from the other two analysed strains than from Chromohalobacter canadensis. That may suggest genomes assignment to two different species of Chromohalobacter. This observation was confirmed by digital DNA-DNA hybridization (dDDH) evaluation, where the similarity between Chromohalobacter 296-RDG and 48-RD10 was 87.5% (d4 method), between 11-W and 296-RDG it was 42.7 % and between 11-W and 48-RD10 it reached 42.9%. Halomonas strains 11-S5 and 25-S5 were clustered together on the whole-genome tree. Allocation to the same species was confirmed by dDDH which was 89.7%. The closest related species to the analysed strains was Halomonas ventosae.
Genome features
| Chromohalobacter 11-W | Chromohalobacter 296-RDG | Chromohalobacter 48-RD10 | Halomonas 11-S5 | Halomonas 25-S5 | |
|---|---|---|---|---|---|
| Genome length (bp) | 3 660 465 | 3 763 726 | 3 785 492 | 3 634 207 | 3 872 356 |
| Number of contigs | 38 | 321 | 222 | 65 | 101 |
| Largest contig (bp) | 607 581 | 256 035 | 627 765 | 320 470 | 407 008 |
| GC content (%) | 60.83 | 60.11 | 60.3 | 66.46 | 65.9 |
| N50 (bp) | 318 640 | 58 943 | 228 913 | 105 985 | 122 550 |
| Number of CDSs | 3 360 | 3 576 | 3 572 | 3 357 | 3 551 |
| Number of rRNAs | 6 | 3 | 3 | 7 | 5 |
| Number of tRNAs | 63 | 63 | 65 | 60 | 58 |
| Number of repeat regions | 3 | 2 | 3 | 1 | 2 |
| Completeness (%) | 98.71 | 98.71 | 99.57 | 99.86 | 99.86 |
| Contamination (%) | 0.86 | 0.86 | 8.42 | 0.68 | 0.54 |
Phylogenetic tree prepared based on 16S rRNA gene sequences analysis using the Neighbor-Joining method and showing the relationships between analysed strains and other Chromohalobacter and Halomonas strains. The evolutionary distances were computed using the Kimura 2-parameter method.
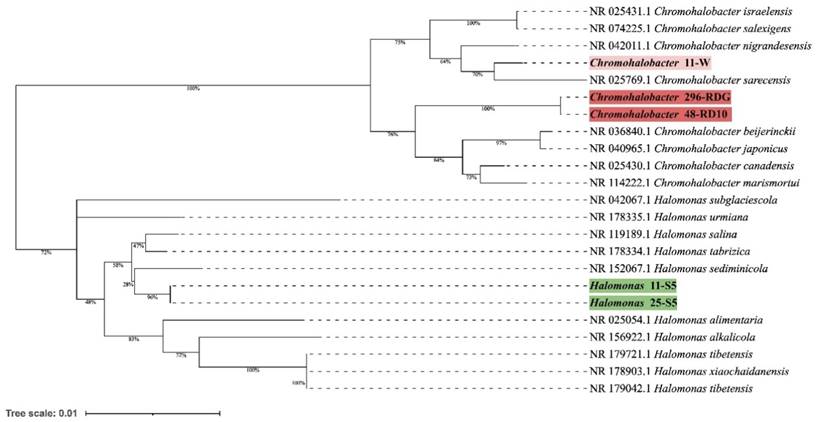
Whole-genome sequence-based phylogenetic tree build using Genome BLAST Distance Phylogeny approach (GBDP) on the Type (Strain) Genome Server (TYGS) platform showing the phylogenetic relationships between analysed Bacteria strains and other close related to them species.
Functional annotation of genomes revealed that they all contained numerous genes involved in the biosynthesis of secondary metabolites (Table 2). However, both Halomonas strains, 11-S5 and 25-S5, had a higher number of genes belonging to this category (99 and 101 genes, respectively) than isolates belonging to the genus Chromohalobacter, which consisted of 69-89 such genes, depending on the strain. This observation is consistent with previous reports that Halomonas and Chromohalobacter have a high diversity of biosynthetic processes (10,12,13) Based on these results, the annotation of BGCs with antiSMASH was performed. Interestingly, as a result, more BGCs were identified in Chromohalobacter strains than Halomonas, despite a smaller number of genes associated with processes identified during the analysis. Moreover, BGCs related to ectoine production have been identified in all genomes. Most likely, it is related to the adaptation of the studied microorganisms to conditions of high salinity. Ectoine produced by the analysed strains is one of the most important compatible solutes that protects the cell against high osmotic pressure (26). In the 11-W, 296-RDG, 48-RD10, and 11-S5 strains, a complete operon ectABC was identified. In the 25-S5 strain, only the ectC gene, essential for ectoine production, was identified. In Halomonas isolates, BGCs associated with ectoine production were the only BGCs identified in the genomes. In Chromohalobacter strains, BGCs related to the production of siderophores, redox-cofactors, and arylopolyenes, were also identified. Table 3 summarizes the information on the identified BGCs in each of the strains.
To summarize, the draft genomes of three Chromohalobacter strains and two Halomonas strains expand the genomic representation in the tree of life. The strains analysed were isolated from the hitherto unexplored saline environment, which allows a deeper understanding of their biodiversity.
eggNOG categories of coding proteins
| Class | Description | Chromohalobacter 11-W [%] | Chromohalobacter 296-RDG [%] | Chromohalobacter 48-RD10 [%] | Halomonas 11-S5 [%] | Halomonas 25-S5 [%] |
|---|---|---|---|---|---|---|
| Information storage and processing | ||||||
| J | Translation, ribosomal structure, and biogenesis | 187 [5.85] | 184 [5.77] | 193 [5.91] | 196 [6.14] | 194 [5.83] |
| A | RNA processing and modification | 0 | 0 | 0 | 0 | 0 |
| K | Transcription | 265 [8.29] | 259 [8.12] | 269 [8.23] | 223 [6.99] | 238 [7.15] |
| L | Replication, recombination, and repair | 129 [4.04] | 187 [5.86] | 211 [6.46] | 178 [5.58] | 232 [6.97] |
| B | Chromatin structure and dynamics | 2 [0.06] | 1 [0.03] | 1 [0.03] | 4 [0.13] | 4 [0.12] |
| Cellular processes and signalling | ||||||
| D | Cell cycle control, cell division, chromosome partitioning | 50 [1.56] | 47 [1.47] | 52 [1.59] | 48 [1.50] | 50 [1.50] |
| Y | Nuclear structure | 0 | 0 | 0 | 0 | 0 |
| V | Defence mechanisms | 34 [1.06] | 41 [1.28] | 34 [1.04] | 50 [1.57] | 53 [1.59] |
| T | Signal transduction mechanisms | 135 [4.23] | 119 [3.73] | 124 [3.80] | 156 [4.89] | 155 [4.66] |
| M | Cell wall/membrane/envelope biogenesis | 201 [6.29] | 227 [7.11] | 240 [7.35] | 171 [5.36] | 176 [5.29] |
| N | Cell motility | 81 [2.54] | 80 [2.51] | 86 [2.63] | 64 [2.01] | 62 [1.86] |
| Z | Cytoskeleton | 0 | 0 | 0 | 0 | 0 |
| W | Extracellular structures | 0 | 0 | 0 | 0 | 0 |
| U | Intracellular trafficking, secretion, and vesicular transport | 69 [2.16] | 76 [2.38] | 77 [2.36] | 61 [1.91] | 60 [1.80] |
| O | Posttranslational modification, protein turnover, chaperones | 114 [3.57] | 108 [3.38] | 119 [3.64] | 138 [4.33] | 137 [4.12] |
| Metabolism | ||||||
| C | Energy production and conversion | 234 [7.32] | 203 [6.36] | 213 [6.52] | 239 [7.49] | 250 [7.51] |
| G | Carbohydrate transport and metabolism | 223 [6.98] | 224 [7.02] | 219 [6.70] | 169 [5.30] | 171 [5.14] |
| E | Amino acid transport and metabolism | 337 [10.55] | 341 [10.69] | 330 [10.10] | 310 [9.72] | 308 [9.26] |
| F | Nucleotide transport and metabolism | 91 [2.85] | 88 [2.76] | 88 [2.69] | 89 [2.79] | 92 [2.77] |
| H | Coenzyme transport and metabolism | 148 [4.63] | 151 [4.73] | 158 [4.84] | 149 [4.67] | 151 [4.54] |
| I | Lipid transport and metabolism | 119 [3.72] | 108 [3.38] | 107 [3.28] | 135 [4.23] | 132 [3.97] |
| P | Inorganic ion transport and metabolism | 231 [7.23] | 224 [7.02] | 213 [6.52] | 233 [7.30] | 237 [7.12] |
| Q | Secondary metabolites biosynthesis, transport, and catabolism | 89 [2.79] | 73 [2.29] | 69 [2.11] | 99 [3.10] | 101 [3.04] |
| Poorly characterized | ||||||
| R | General function prediction only | 0 | 0 | 0 | 0 | 0 |
| S | Function unknown | 618 [19.34] | 614 [19.24] | 626 [19.16] | 616 [19.31] | 646 [19.42] |
| All proteins | 3195 | 3191 | 3267 | 3190 | 3327 | |
BGCs identified with AntiSMASH in the analysed genomes
| Sample name | Chromohalobacter 11-W | Chromohalobacter 296-RDG | Chromohalobacter 48-RD10 | Halomonas 11-S5 | Halomonas 25-S5 |
|---|---|---|---|---|---|
| AntiSMASH | |||||
| # of BGC | 5 | 7 | 4 | 1 | 1 |
| Arylpolyene | 1 | 1 | 1 | 0 | 0 |
| Betalactone | 1 | 1 | 0 | 0 | 0 |
| Butyrolactone | 0 | 1 | 0 | 0 | 0 |
| Ectoine | 1 | 1 | 1 | 1 | 1 |
| Siderophore | 1 | 1 | 1 | 0 | 0 |
| Phosphonate | 0 | 1 | 0 | 0 | 0 |
| Redox-cofactor | 1 | 1 | 1 | 0 | 0 |
Acknowledgements
This research was financed from the following funds: "InterDOC-STARt" project (POWR.03.02.00-00-I033/16-00), the MINI InterDOC-STARt grant and by the subsidy for scientific activity provided to the Department of Molecular Microbiology by the University of Lodz (B2111000000038.01). The authors are grateful to the management of the Bochnia Salt Mine for granting permission for sampling, Mr Tomasz Migdas, the Technical Director of the Bochnia Salt Mine, for his invaluable help in organizing sampling and for inspiring conversation, and to the mine geologists: Michał Flasza and Aleksandra Puławska for guiding the sampling tours. We would also like to thank Dr Magdalena Kowalewicz-Kulbat and Dr Dominika Drzewiecka for preparing pure strain cultures and Professor Łukasz Dziewit for a fruitful discussion and helpful advice regarding this publication.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Yin J, Chen JC, Wu Q, Chen GQ. Halophiles, coming stars for industrial biotechnology. Biotechnol Adv [Internet]. 2015;33(7):1433-42 Available from: http://dx.doi.org/10.1016/j.biotechadv.2014.10.008
2. Shang J, Xu L, Yang R, Zhao Y, Tang S, Sun J. Halomonas alkalisoli sp. nov, a novel haloalkalophilic species from saline-alkaline soil, and reclassification of Halomonas daqingensis Wu et al. 2008 as a later heterotypic synonym of Halomonas desiderata. Syst Appl Microbiol [Internet]. 2022;45(5):126351. Available from: https://doi.org/10.1016/j.syapm.2022.126351
3. Gong Q, Zhao P, Miao S, Yi K, Ma C, Zhang G. Halorubrum salipaludis sp. nov, isolated from the saline-alkaline soil. Arch Microbiol [Internet]. 2022;204(1):1-6 Available from: https://doi.org/10.1007/s00203-021-02729-1
4. Zuo Z, Zhao D, Zhou J, Han J, Xiang H. Halalkalirubrum salinum gen. nov, sp. nov, a halophilic archaeon isolated from a saline lake. Antonie van Leeuwenhoek, Int J Gen Mol Microbiol [Internet]. 2021;114(1):83-94 Available from: https://doi.org/10.1007/s10482-020-01502-6
5. Sun S, Chen F, Xu Y, Liu J, Chen S. Halorubrum amylolyticum sp. nov, a novel halophilic archaeon isolated from a salt mine. Antonie van Leeuwenhoek, Int J Gen Mol Microbiol [Internet]. 2019;112(12):1849-61 Available from: https://doi.org/10.1007/s10482-019-01313-4
6. Lülf RH, Hilgarth M, Ehrmann MA. Chromohalobacter moromii sp. nov, a moderately halophilic bacterium isolated from lupine-based moromi fermentation. Syst Appl Microbiol. 2022 45(4)
7. Ventosa A, Gutierrez MC, Garcia MT, Ruiz-Berraquero F. Classification of “Chromobacterium marismortui” in a new genus, Chromohalobacter gen. nov, as Chromohalobacter marismortui comb. nov, nom. rev. Int J Syst Bacteriol. 1989;39(4):382-6
8. Vreeland RH, Litchfield CD, Martin EL, Elliot E. Halomonas elongata, a new genus and species of extremely salt-tolerant bacteria. Int J Syst Bacteriol. 1980;30(2):485-95
9. Parker CT, Tindall BJ, Garrity GM. International code of nomenclature of Prokaryotes. Int J Syst Evol Microbiol. 2019;69(1):S1
10. Argandoña M, Piubeli F, Reina-Bueno M, Nieto JJ, Vargas C. New insights into hydroxyectoine synthesis and its transcriptional regulation in the broad-salt growing halophilic bacterium Chromohalobacter salexigens. Microb Biotechnol. 2021;14(4):1472-93
11. Navarro-Torre S, Carro L, Rodríguez-Llorente ID, Pajuelo E, Caviedes MÁ, Igual JM. et al. Halomonas radicis sp. nov, isolated from Arthrocnemum macrostachyum growing in the Odiel marshes(Spain) and emended descriptions of Halomonas xinjiangensis and Halomonas zincidurans. Int J Syst Evol Microbiol. 2020;70(1):220-7
12. Poli A, Nicolaus B, Denizci AA, Yavuzturk B, Kazan D. Halomonas smyrnensis sp. nov, a moderately halophilic, exopolysaccharide-producing bacterium. Int J Syst Evol Microbiol. 2013;63(1):10-8
13. Thomas T, Elain A, Bazire A, Bruzaud S. Complete genome sequence of the halophilic PHA-producing bacterium Halomonas sp. SF2003: insights into its biotechnological potential. World J Microbiol Biotechnol [Internet]. 2019;35(3):1-14 Available from: http://dx.doi.org/10.1007/s11274-019-2627-8
14. Andrews S. FastQC: a quality control tool for high throughput sequence data [Internet]. 2022. Available from: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
15. Krueger F. Trim Galore [Internet]. 2022. Available from: https://www.bioinformatics.babraham.ac.uk/projects/trim_galore/.
16. Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS. et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19(5):455-77
17. Parks DH, Imelfort M, Skennerton CT, Hugenholtz P, Tyson GW. CheckM: Assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 2015;25(7):1043-55
18. Mikheenko A, Prjibelski A, Saveliev V, Antipov D, Gurevich A. Versatile genome assembly evaluation with QUAST-LG. Bioinformatics. 2018;34(13):i142-50
19. Seemann T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics. 2014;30(14):2068-9
20. Huerta-Cepas J, Szklarczyk D, Heller D, Hernández-Plaza A, Forslund SK, Cook H. et al. EggNOG 5.0: A hierarchical, functionally and phylogenetically annotated orthology resource based on 5090 organisms and 2502 viruses. Nucleic Acids Res. 2019;47(D1):D309-14
21. Blin K, Shaw S, Kloosterman AM, Charlop-Powers Z, Van Wezel GP, Medema MH. et al. AntiSMASH 6.0: Improving cluster detection and comparison capabilities. Nucleic Acids Res. 2021;49(W1):W29-35
22. Chaumeil PA, Mussig AJ, Hugenholtz P, Parks DH. GTDB-Tk: A toolkit to classify genomes with the genome taxonomy database. Bioinformatics. 2020;36(6):1925-7
23. Meier-Kolthoff JP, Göker M. TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat Commun [Internet]. 2019 10(1). Available from: http://dx.doi.org/10.1038/s41467-019-10210-3
24. Letunic I, Bork P. Interactive tree of life (iTOL) v5: An online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021;49(W1):W293-6
25. Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. 2018;35(6):1547-9
26. Czech L, Hermann L, Stöveken N, Richter AA, Höppner A, Smits SHJ. et al. Role of the extremolytes ectoine and hydroxyectoine as stress protectants and nutrients: Genetics, phylogenomics, biochemistry, and structural analysis. Genes (Basel). 2018;9(4):1-58
Author contact
Corresponding authors: Jakub Lach and Paweł Stączek: jakub.lachuni.lodz.pl, pawel.staczekuni.lodz.pl

 Global reach, higher impact
Global reach, higher impact