ISSN: 1839-9940
J Genomics 2019; 7:50-55. doi:10.7150/jgen.35875 This volume Cite
Short Research Paper
Draft genome sequences for three unisolated Alnus-infective Frankia Sp+ strains, AgTrS, AiOr and AvVan, the first sequenced Frankia strains able to sporulate in-planta
1. Univ Lyon, Université Lyon 1, CNRS, UMR5557, Ecologie Microbienne, INRA, UMR 1418, 43 bd du 11 novembre 1918, F-69622 Villeurbanne, France
2. Univ Lyon, Université Lyon 1, CNRS, UMR5558, Laboratoire de Biométrie et Biologie Évolutive, 43 bd du 11 novembre 1918, F-69622 Villeurbanne, France
3. Univ Lyon, Université Lyon 1, DTAMB, FR 3728 BioEnviS, 43 bd du 11 novembre 1918, F-69622 Villeurbanne, France
4. Biofidal, 170 av Gabriel Péri, F-69518 Vaulx-en-Velin, France
Received 2019-4-19; Accepted 2019-7-22; Published 2019-9-17
Abstract
Actinobacteria from genus Frankia are able to form symbiotic associations with actinorhizal plants including alders. Among them, Sp+ strains are characterized by their ability to differentiate numerous sporangia inside host plant cells (unlike “Sp-” strains unable of in-planta sporulation). Here, we report the first genome sequences of three unisolated Sp+ strains: AgTrS, AiOr and AvVan obtained from Alnus glutinosa, A. incana and A. alnobetula (previously known as viridis), respectively (with genome completeness estimated at more than 98%). They represent new Frankia species based on Average Nucleotide Identity (ANI) calculations, and the smallest Alnus-infective Frankia genomes so far sequenced (~5 Mbp), with 5,178, 6,192 and 5,751 candidate protein-encoding genes for AgTrS, AiOr and AvVan, respectively.
Keywords: Frankia, AgTrS, AiOr, AvVan
Genome Announcement
Frankia strains are filamentous actinobacteria able to fix nitrogen and to form symbiotic associations with actinorhizal plants, leading to the formation of root nodules where trophic exchanges between plant and bacteria take place. Phylogenetic studies showed that clades within Frankia genus are strongly linked to infection groups, with Cluster 1 containing strains infective on Alnus and Myrica [1, 2]. Frankia is also characterized by its ability to differentiate sporangia. Most isolated Frankia strains have been described as sporulating in-vitro [1, 3]. However, certain strains, called “Sp+”, have the ability to sporulate inside host cells (unlike “Sp-” strains unable of in-planta sporulation) [4]. Sp+ strains have been commonly reported in association with alders, especially A. glutinosa, A. incana and A. alnobetula (formerly A. viridis) species. In contrast to Sp- strains, up to date, Sp+ strains are still totally culture recalcitrant (none are available in pure culture despite many isolation attempts) [5]. Furthermore, we recently described their narrower host specificity [6], suggesting a strong host dependence. It was hypothesized that Sp+ strains could have evolved towards an obligatory symbiont status with spores representing their only form of survival outside the host. Indeed, produced early and abundantly in host cells, spores would be released during nodule senescence, thus enabling Sp+ strains to survive and disseminate in the soil in a free state [7]. Recently, MLSA-based studies directly conducted on Sp+ nodules collected from various geographical sites confirmed that Alnus-infective Sp+ strains belonged to Cluster 1 as expected. These studies also showed that the Sp+ trait was associated with distinct phylogenetic lineages, strongly correlated to the host species [7, 2], suggesting that Sp+ strains had emerged several times independently over the course of Frankia diversification. To date, more than thirty Frankia strains covering the diversity of the Frankia genus have been sequenced [8], helping to predict and identify pathways involved in the biosynthesis of natural products by Frankia [9, 10]. However, no Frankia Sp+ genomes have been reported so far. Here, we reported the sequencing of three Sp+ Frankia genomes. The main challenge was to get DNA of these unisolated strains directly from nodules, limiting plant DNA contaminations. For this, we optimized a protocol of DNA extraction from Frankia spores directly isolated from nodules.
Three Alnus-infective Sp+ Frankia uncultured strains from Cluster 1, AgTrS, AiOr and AvVan, were selected from nodules collected in 3 distinct alder stands, colonized by A. glutinosa (Le Tremblay, Savoie, France), A. incana (Ornon, Isère, France) and A. alnobetula (Vanoise, Savoie, France), respectively [6]. AgTrS, AiOr and AvVan genomes were sequenced using DNA extracted from spores directly isolated from nodules. For each strain, at least 1 g of surface-sterilized (with calcium hypochlorite 1 % w/v, 15 minutes) and peeled nodules was crushed in liquid nitrogen, with 10 mL of buffer containing 0.5 M Tris-HCl pH 7, 4 % PVP (w/v), 0.1 M KCl, 5 mM EDTA, 0.6 M sucrose, 10 mM Na2S2O3. Crushed nodule suspensions were successively filtered through 100 µM and 20 µM sterile-filters (Steriflip® Filter Millipore, Life Science-Merck, Paris, France) to separate Frankia spores from plant residues and Frankia hyphae and vesicules. Frankia spore suspensions were homogenized using TissueLyser II, (Qiagen, Courtaboeuf, France) for 40 sec. at 20 Hz before total DNA extraction. DNA was extracted with FastDNA® SPIN for Soil kit (MP Biomedicals, Illkirch, France) following the supplied procedure. DNA samples were shotgun sequenced after a Nextera XT library construction step (Illumina, USA), using Illumina MiSeq technology with a paired-end 2 × 300-bp run (MiSeq 600 cycles V3 kit, Biofidal, Lyon, France). It is worth noting that an additional sequencing was conducted on AgTrS strain using 454-pyrosequencing technology (Life Sciences- Merck), however this did not allow to improve genome assembly and was thus not performed for the two other strains AiOr and AvVan. Genome assemblies were realised using Unicycler v0.4.3 [11] and their annotation was done with the MicroScope platform version 3.10.0 [12].
A total of 3,480,805 reads were generated for AgTrs, 4,413,305 reads for AiOr and 1,805,928 reads for AvVan. Reads were sorted by nucleotide frequencies using Perl scripts to remove the reads with G+C content ≤ 54 %, since they are likely due to host plant DNA contaminations. More precisely, this threshold was based on the high G+C content reported in Frankia genomes [13], with a 72% overall G+C content (only 26 short genes below 54% GC and a single group of 5 very short genes below 54% G+C), against a mean G+C content of alder genomes of ~40% [14]. Based on G+C content read sorting, a final set of 2,401,363 reads was retained for AgTrS, 3,977,168 reads for AiOr and 549,771 reads for AvVan. Seventy-six to 96% of eliminated reads from AgTrS, AiOr and AvVan sequencing data showed percent sequence identity ID > 85 % against A. glutinosa genome (accession no. ASM325496v1) and less than 1% against Frankia genomes on MicroScope platform (only 0.1 and 0.2% for AgTrS and AvVan, respectively). Genome assemblies based on sorted reads showed a reduced number of contigs as well as an increased mean contig size compared to assemblies based on unsorted reads, suggesting a significant improvement of genome assemblies (Table 1).
Assembly data of the three Sp+ Frankia genomes sequenced, before and after read sorting based on their G+C content (to remove the reads with G+C content ≤ 54 %).
| Data before read sorting | Data after read sorting | |||||
|---|---|---|---|---|---|---|
| Sp+ strain | Read number | Total contig number (with ≥ 5 kb) | Mean contig size (pb) | Read number | Total contig number (with ≥ 5 kb) | Mean contig size (pb) |
| AgTrS | 3,480,805 | 3,559 (366) | 2,120 | 2,401,363 (69%) | 612 (281) | 7,978 |
| AiOr | 4,413 305 | 1,860 (376) | 4,542 | 3,977,168 (90%) | 669 (302) | 8,228 |
| AvVan | 1,805,928 | 7,798 (457) | 2,876 | 549,771 (30%) | 1,228 (322) | 3,962 |
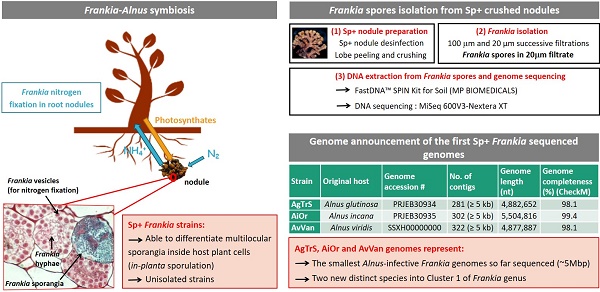
Assembly data are summarized in Table 2 together with genomes associated with Frankia species, already described or soon to be. The final draft assembly for AgTrS consisted of 281 contigs (≥ 5 kb). The maximum length and N50 values of the contigs were 96.9 kb and 15.3 kb, respectively, giving a total genome size of 4,882,652 bp. For AiOr, the final draft assembly consisted of 302 contigs (≥ 5 kp) containing 5,504,816 bp, with a maximum contig length of 105.2 kb and a N50 value of 17.4 kb. Both AgTrS and AiOr draft genomes had an overall G+C content of 71.6%. For AvVan, the final draft assembly consisted of 322 contigs (≥ 5 kb), with the contig maximum length and N50 values of 30.1 kb and 6.6 kb, respectively. It contained a total sequence of 4,877,887 bp, with an overall G+C content of 71.4%. Genome completeness was estimated at 98.1% for AgTrS and AvVan strains and 99.4% for AiOr, using CheckM software that assesses the presence of a specific number of markers depending on the studied organism (307 markers for Frankia genomes) [15]. The assembled genomes of AgTrS, AiOr and AvVan strains resulted in 5,178, 6,192 and 5,751 candidate protein-encoding genes, respectively (Table 2). Classification of proteins into their COG functional categories (using MicroScope Platform from Genoscope, http://www.genoscope.cns.fr/agc/microscope/home/index.php) showed similar proportions of proteins in the different functional groups among the three strains (Figure 1).
Genome features of the three Sp+ Frankia genomes sequenced (in grey), compared to available Frankia genomes (only genomes associated with described or being described Frankia species were included in this table and they are all Sp- strains).
| Species | Cluster | Strain | Genome accession # | No. of contigs | Genome length (nt) | Genomic G+C content (mol%) | N50 genomic values | Genome coverage | Genome completeness (%) (CheckM) | Original host genus | Associated hosts | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| - | 1 | AgTrS | PRJEB30934 | 281 (≥ 5 kb) | 4,882,652 | 71.6 | 15.3 | 295.1 | 98.1 | Alnus | A glutinosa, A. incana | This study, [6] |
| - | 1 | AiOr | PRJEB30935 | 302 (≥ 5 kb) | 5,504,816 | 71.6 | 17.4 | 433.5 | 99.4 | Alnus | A. incana | This study, [6] |
| - | 1 | AvVan | SSXH00000000 | 322 (≥ 5 kb) | 4,877,887 | 71.4 | 6.6 | 67.6 | 98.1 | Alnus | A. viridis | This study, [6] |
| F. alni | 1 | DSM 45986T (ACN14aT) | NC_008278.1 | 1 | 7,497,934 | 72.8 | - | - | - | Alnus | Alnus, Myricaceae | [13] |
| F. torreyi | 1 | DSM 44263T (CpI1-ST) | JYFN00000000 | 153 | 7,624,758 | 72.4 | - | - | - | Comptonia | Alnus, Myricaceae | [19] |
| F. canadensis | 1 | DSM 45898T (ARgP5T) | OESX01000001 | 568 | 7,730,285 | 72.4 | - | - | - | Alnus | Alnus, Myricaceae | [20] |
| F. casuarinae | 1 | DSM 45818T (CcI3T) | CP000249.1 | 1 | 5,433,628 | 70.1 | - | - | - | Casuarina | Casuarinaceae (except Gymnostoma) | [13] |
| F. coriariae | 2 | DSM 100624T (BMG5.1T) | JWIO00000000 | 116 | 5,795,263 | 71.0 | - | - | - | Coriaria | Datisca, Coriaria | [21] |
| Candidatus F. californiensis | 2 | Dg2 | FLUV00000000 | 1066 | 5,929,312 | 67.9 | - | - | - | Datisca | Rosaceae, Datisca | [22] |
| Candidatus F. datiscae | 2 | Dg1 | CP002801 | 1 | 5,323,186 | 70.0 | - | - | - | Datisca | Datisca, Coriaria | [23] |
| F. discariae | 3 | DSM 46785T (BCU110501T) | ARDT00000000 | 207 | 7,891,711 | 72.3 | - | - | - | Discaria | Rhamnaceae, Elaeagnaceae, Gymnostoma | [24] |
| F. elaeagni | 3 | DSM 46783T (BMG5.12T) | ARFH00000000 | 139 | 7,589,313 | 71.7 | - | - | - | Elaeagnus | Rhamnaceae, Elaeagnaceae, Gymnostoma | [25] |
| F. irregularis | 3 | DSM 45899T (G2T) | FAOZ00000000 | 83 | 9,537,992 | 70.9 | - | - | - | Casuarina | Rhamnaceae, Elaeagnaceae, Gymnostoma | [26] |
| F. inefficax | 4 | DSM 45817T (EuI1cT) | CP002299.1 | 1 | 8,815,781 | 72.3 | - | - | - | Elaeagnus | Elaeagnaceae, Morella | [8] |
| F. saprophytica | 4 | DSM 105290T (Cn3T) | AGJN00000000 | 2 | 9,978,592 | 71.8 | - | - | - | Coriaria | - | [27] |
| F. asymbiotica | 4 | DSM 100626T (M16386T) | MOMC00000000.1 | 174 | 9,435,764 | 72.0 | - | - | - | Morella | - | [28] |
COG functional classification of proteins encoded on the three sequenced Sp+ Frankia genomes AgTrS, AiOr and AvVan. Proportions (%) of proteins in each of the COG super-functional categories “Cellular processes and signalling”, “Information processing and storage”, “Metabolism” and “Poorly characterized”, predicted for AgTrS (white bars), AiOr (black bars) and AvVan (grey bars) genomes.
Frankia sp. AgTrS, AiOr and AvVan Sp+ strains represent the smallest Alnus-infective Frankia genomes so far sequenced (~5 Mbp), close to the genome size of Casuarina-infective strains previously described as subservient to their host [16]. In order to place the three Sp+ strains in Frankia reference phylogeny and to assess the relationships between them, a maximum likelihood phylogeny was inferred (Figure 2). More precisely, the 28 Frankia genomes available from NCBI were retrieved (for all these strains, origins and genome features have been summarized by Tisa et al. [8]) and gathered in a local database together with the 3 Sp+ assemblies. This dataset included seven strains from Cluster 1 unable to sporulate in-planta, thus Sp- strains: ACN14a as F. alni species representative, AvcI1, ACN1ag, CpI1P, CpI1S, QA3 and ARgP5. Fifty-one ribosomal proteins were retrieved from the 31 genomes and combined to build a large supermatrix of (18,582 nucleotide positions) that was used for phylogenetic inferences. The ML tree was built with IQ TREE [17] with the GTR+I+R4 evolutionary model as suggested by the model selection tool implemented in IQ TREE. The branch robustness of the ML tree was estimated with the non-parametric bootstrap procedure Implemented in IQ TREE (100 replicates of the original alignment). The resulting tree confirmed the position of the 3 Sp+ Frankia strains AgTrS, AiOr and AvVan into Cluster 1 (Figure 2). In this cluster, AvVan and AiOr appeared closely related to ACN14a, AvcI1, ACN1ag, Cpl1P, Cpl1S, and QA3 strains (bootstrap value = 100%), while AgTrS formed a distinct lineage (Figure 2), suggesting that the three Sp+ strains belonged to two different clades as previously discussed [2, 7].
Position of the three Sp+ sequenced Frankia strains AgTrS, AiOr and AvVan in Frankia genus phylogeny based on ribosomal proteins. In addition to Sp+ genomes, a total of 28 sequenced Frankia strains were used. For all the 31 genomes, 51 ribosomal protein sequences (total size = 18,582 nt) were included in a supermatrix and the phylogenetic tree was constructed based on the model GTR+I+R4.
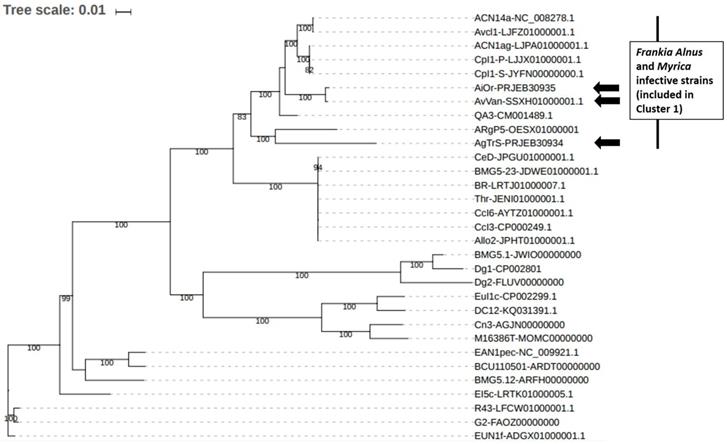
Average Nucleotide Identity (ANI) calculations were performed in order to accurately distinguish between strains at the species level into the Cluster 1, using the recommended cut-off point of 95 % ANI for species delineation [18]. All 3 Sp+ Frankia genomes AgTrS, AiOr, and AvVan showed less than 90.1% similarity with the genomes of ACN14a and QA3 Alnus-infective Frankia strains from Cluster 1 (both ACN14a and QA3 have also been included in the phylogenetic tree in Figure 2). Only two genomes, AvVan and AiOr shared 98.5% ANI, which is above the threshold value for species circumscription. These phylogenomic analyses confirm the results obtained by a large survey on Sp+ strains that showed the genetic divergence between A. glutinosa-infective strains and A. alnobetula- and A. incana-infective strains [2, 7]. These results lead to conclude that AgTrS, AiOr and AvVan most likely represent two new distinct species into Cluster 1 of Frankia genus, with AiOr and AvVan belonging to the same species.
In conclusion, the genome sequencing of the three Frankia Sp+ strains AgTrS, AiOr and AvVan offer a unique opportunity to explore the evolution of their life history traits. Thorough analyses based on comparative genomic approaches with Frankia Sp- genomes already available will be performed, for instance to look for clues to Sp+ strain ability to sporulate in-planta, to their non-cultivability/host dependence, to their higher narrower host specificity, and eventually clarify their hypothetical status of obligatory symbiont.
Nucleotide sequence accession numbers
This whole-genome shotgun project has been deposited in DDBJ/EMBL/GenBank under the accession no. PRJEB30934, PRJEB30935 and SSXH00000000 (for Frankia sp. AgTrS, AiOr and AvVan). The version described in this paper is the first version. No pure culture of AgTrS, AiOr and AvVan strains are available, these strains are maintained in the UMR5557 Microbial Ecology of Lyon (France) on Alnus seedlings (under controlled hydroponic conditions) and they are available as nodules to the research community upon request.
Acknowledgements
Partial funding was provided by the National Center for Scientific Research (CNRS, grant EC2CO) and the research cluster FR41 BioEnviS. Thanks are expressed to Roxane Bai and Cornelia Alvarez (University Claude Bernard, Lyon1) for technical assistance, as well as Laetitia Cotin-Galvan, Adrien Pozzi and Guillaume Schwob (University Claude Bernard, Lyon1) for help with nodule samplings. We also thank the Genoscope (the French National Sequencing Center, Evry) for help with genome annotations.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Normand P, Orso S, Cournoyer B, Jeannin P, Chapelon C. et al. Molecular phylogeny of the genus Frankia and related genera and emendation of the family Frankiaceae. Int J Syst Bacteriol. 1996;46:1-9
2. Pozzi AC, Bautista-Guerrero HH, Abby SS, Herrera-Belaroussi A, Abrouk D. et al. Multi-Locus Sequence Analysis and extensive sampling bring new insights on Frankia phylogeny and phylogeography. Syst. Appl. Microbiol. 2018 S0723-2020(18)30113-9
3. Callaham D, Del Tredici P, Torrey JG. Isolation and cultivation in vitro of the actinomycete causing root nodulation in Comptonia. Science. 1978;199:899-902
4. van Dijk C. Spore formation and endophyte diversity in root nodules of Alnus glutinosa (L.) Vill. New Phytol. 1978;81:601-615
5. Torrey J. Endophyte sporulation in root nodules of actinorhizal plants. Physiol Plant. 1987;70:279-288
6. Schwob G, Roy M, Pozzi AC, Herrera-Belaroussi A, Fernandez MP. In planta sporulation of Frankia spp. as a determinant of alder-symbiont interactions. Appl Environ Microbiol. 2018;84:e01737-01718
7. Pozzi AC, Bautista-Guerrero HH, Nouioui I, Cotin-Galvan L, Pepin R. et al. In-planta sporulation phenotype: a major life history trait to understand the evolution of Alnus-infective Frankia strains. Environ. Microbiol. 2015;17:3125-3138
8. Tisa LS, Oshone R, Sarkar I, Ktari A, Sen A. et al. Genomic approaches toward understanding the actinorhizal symbiosis: An update on the status of Frankia genomes. Symbiosis. 2016;70:5-16
9. Udwary DW, Gontang EA, Jones AC, Jones CS, Schultz AW. et al. Significant natural product biosynthetic potential of actinorhizal symbionts of the genus frankia, as revealed by comparative genomic and proteomic analyses. Appl Environ Microbiol. 2011;77:3617-3625
10. Deicke M, Mohr JF, Roy S, Herzsprung P, Bellenger JP. et al. Metallophore profiling of nitrogen-fixing Frankia spp. to understand metal management in the rhizosphere of actinorhizal plants. Metallomics. 2019;11:810-821
11. Wick R, Judd L, Gorrie C, Holt K. Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput Biol. 2017;13:e1005595
12. Vallenet D, Calteau A, Cruveiller S, Gachet M, Lajus A. et al. MicroScope in 2017: an expanding and evolving integrated resource for community expertise of microbial genomes. Nucleic Acids Res. 2017;45:D517-D528
13. Normand P, Lapierre P, Tisa LS, Gogarten JP, Alloisio N. et al. Genome characteristics of facultatively symbiotic Frankia sp. strains reflect host range and host plant biogeography. Genome Res. 2007;17:7-15
14. Griesmann M, Chang Y, Liu X, Spannagl M, Crook MB. et al. Phylogenomics reveals multiple independent losses of the nitrogen-fixing root nodule symbiosis. Science. 2018;361:6398
15. Parks D, Imelfort M, Skennerton C, Hugenholtz P, Tyson G. CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 2015;25:1043-1055
16. Tisa LS, Beauchemin N, Gtari M, Sen A, Wall LG. What stories can the Frankia genomes start to tell us? J Biosci. 2013;38:719-726
17. Nguyen L, Schmidt H, von Haeseler A, Minh B. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol. 2015;32:268-274
18. Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P. et al. DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol. 2007;57:81-91
19. Oshone R, Hurst SGt, Abebe-Akele F, Simpson S, Morris K. et al. Permanent draft genome sequences for two variants of Frankia sp. strain CpI1, the first Frankia strain isolated from root nodules of Comptonia peregrina. Genome Announc. 2016:4
20. Normand P, Nouioui I, Pujic P, Fournier P, Dubost A. et al. Frankia canadensis sp. nov, isolated from root nodules of Alnus incana subspecies rugosa. Int. J. Syst. Evol. Microbiol. 2018:10 1099/ijsem.0.002939
21. Gtari M, Ghodhbane-Gtari F, Nouioui I, Ktari A, Hezbri K. et al. Cultivating the uncultured: growing the recalcitrant cluster-2 Frankia strains. Sci. Rep. 2015;5:13112
22. Nguyen TV, Wibberg D, Battenberg K, Blom J, Vanden Heuvel B. et al. An assemblage of Frankia Cluster 1I strains from California contains the canonical nod genes and also the sulfotransferase gene nodH. BMC Genomics. 2016;17:796
23. Persson T, Benson DR, Normand P, Vanden Heuvel B, Pujic P. et al. Genome sequence of "Candidatus Frankia datiscae" Dg1, the uncultured microsymbiont from nitrogen-fixing root nodules of the dicot Datisca glomerata. J Bacteriol. 2011;193:7017-7018
24. Wall LG, Beauchemin N, Cantor MN, Chaia E, Chen A. et al. Draft genome sequence of Frankia sp. strain BCU110501, a nitrogen-fixing actinobacterium isolated from nodules of Discaria trinevis. Genome Announc. 2013;1:e00503-00513
25. Nouioui I, Beauchemin N, Cantor MN, Chen A, Detter JC. et al. Draft Genome Sequence of Frankia sp. Strain BMG5.12, a Nitrogen-Fixing Actinobacterium Isolated from Tunisian Soils. Genome Announc. 2013:1
26. Nouioui I, Gtari M, Goker M, Ghodhbane-Gtari F, Tisa LS. et al. Draft Genome Sequence of Frankia Strain G2, a Nitrogen-Fixing Actinobacterium Isolated from Casuarina equisetifolia and Able To Nodulate Actinorhizal Plants of the Order Rhamnales. Genome Announc. 2016:4
27. Ghodhbane-Gtari F, Beauchemin N, Bruce D, Chain P, Chen A. et al. Draft genome sequence of Frankia sp. strain CN3, an atypical, noninfective (Nod-) ineffective (Fix-) isolate from Coriaria nepalensis. Genome Announc. 2013;1:e0008513
28. Nouioui I, Gueddou A, Ghodhbane-Gtari F, Rhode M, Gtari M. et al. Frankia asymbiotica sp. nov, a non-infective actinobacterium isolated from Morella californica root nodule. Int. J. Syst. Evol. Microbiol. 2017 doi: 10.1099
Author contact
Corresponding author: Herrera-Belaroussi Aude, UMR 5557 Ecologie Microbienne 43, Boulevard du 11 novembre 1918, F-69622 Villeurbanne Cedex, France. Tel: (33) 472 448 200 E-mail: aude.herrera-belaroussifr

 Global reach, higher impact
Global reach, higher impact